|
Previous Chapter |
 Contents Page |
Next Chapter |
|
Previous Chapter |
 Contents Page |
Next Chapter |
A feeding trial was conducted to test the response of O. brumata to feeding on Calluna plants grown under a factorial combination of raised CO2 and nitrogen fertiliser treatments. In addition, tissue nitrogen concentrations of field-sampled Calluna plants were determined, in order to test the hypothesis that outbreaks occur on nitrogen-enriched vegetation. Experimental CO2-enrichment had no significant effect on Calluna growth or tissue chemistry, and no significant effect on O. brumata survival or development. There was no evidence for nutrient-limitation of the response of Calluna to CO2 enrichment. Nitrogen fertilisation increased Calluna shoot growth rate and water content, decreased foliar C:N ratio, and increased O. brumata growth rate, development rate and pupal weight. Larval survival rate was positively related to Calluna current year's shoot length at the date of hatching. There was no evidence that Calluna from two O. brumata outbreak sites on Orkney was nitrogen-enriched, compared with control non-outbreak sites on Orkney and mainland Scotland.
The effects of future environmental changes on insect-plant systems are currently subject to much investigation (Harrington & Stork, 1995). Increasing atmospheric carbon dioxide (CO2) concentrations, and increasing deposition of nitrogenous pollutants, both have the potential to alter plant nutritional quality and insect performance (Brown, 1995; Pitcairn et al., 1995; Watt et al., 1995; Docherty et al., 1996). In this chapter, the effects of a factorial combination of increased CO2 concentration and increased nitrogen availability on the O. brumata-C. vulgaris system were tested. In addition, the hypothesis that moorland O. brumata outbreaks are stimulated by local nitrogen-enrichment of C. vulgaris (see Section 1.2.3) was tested by chemical analysis of field-sampled vegetation.
Global atmospheric CO2 concentrations are predicted to double in the next century (Houghton et al., 1992; Vitousek, 1992), and the potential effects of this change on insect herbivory have received much recent attention (see Section 1.6 for a review). Experimental CO2-enrichment usually produces either negative effects or no significant effect on plant nitrogen concentration and insect development (Watt et al., 1995). Studies of O. brumata development on sycamore (Acer pseudoplatanus) and beech (Fagus sylvatica) have found no significant effect of CO2-enrichment, whilst similar studies of O. brumata on Sitka spruce (Picea sitchensis) showed reduced larval growth rate, but no change in consumption or development time, on CO2-enriched plants (Watt et al., 1996).
The objective of this study was to test the effect of CO2-enrichment on the O. brumata-C. vulgaris system. A feeding trial was conducted, in which O. brumata larvae were reared upon Calluna plants growing under ambient or raised CO2 atmospheres in open-top fumigation chambers. Since it has been proposed that the response of Calluna to CO2-enrichment is nutrient-limited (Woodin et al., 1992; but see Section 1.6), the effects of CO2 enrichment were studied under both low and high nitrogen treatments, using a factorial experimental design. Because long-term responses to CO2 can differ from short-term effects (Woodin et al., 1992), this study ran continuously for 20 months, spanning two O. brumata feeding seasons.
Whilst CO2 and nitrogen availability have direct effects on host quality and insect nutrition (Section 1.6), they can also cause more subtle changes in host phenology; for example, Sitka spruce budburst date is delayed by CO2-enrichment (Murray et al., 1994). Changes in host phenology can have repercussions for insect herbivory, particularly if hatch-budburst synchrony is important for larval establishment (Dewar & Watt, 1992) (but see Chapter 5). In the present study, Calluna shoot lengths were monitored in order to detect any effects of the CO2 and nitrogen treatments on growth phenology; the importance of Calluna phenology for O. brumata development, particularly during the early larval stages, is discussed.
Chapter 1 reviewed the hypothesis that insect outbreaks are caused by host nitrogen-enrichment. In the case of Calluna, there is good evidence that tissue nitrogen concentrations are increasing in Britain, in response to the national pattern of increasing nitrogen deposition (Pitcairn et al., 1995), and there is evidence from studies of Lochmaea suturalis in Holland that such nitrogen enrichment can stimulate insect outbreaks (Berdowski, 1993) (Section 2.3.3). The hypothesis that the same mechanism is responsible for stimulating the recent moorland O. brumata outbreaks in Scotland was introduced in Section 1.2.3. This hypothesis is strengthened by the result of Chapter 6, that nitrogen fertilisation of Calluna seedlings resulted in increased O. brumata pupal weight, but is weakened by the insensitivity of O. brumata to a natural decline in Calluna nitrogen concentration over time, shown in Chapter 5.
In this chapter, the use of a nitrogen fertiliser treatment in the feeding trial described above (Section 7.1.2) permitted a further test of the hypothesis that Calluna nitrogen-enrichment improves O. brumata performance; in this case, the insect responses were complemented by Calluna tissue nitrogen data (which were lacking in a previous test of this hypothesis; see Chapter 6). In addition, a more direct test for nitrogen enrichment of Calluna was made in the field: Calluna was sampled from O. brumata outbreak and non-outbreak areas, and its tissue nitrogen concentration determined; significantly higher nitrogen concentrations in Calluna from outbreak areas than from non-outbreak areas would support the hypothesis that outbreaks are triggered by host nitrogen enrichment.
-- fumigation system
Eight open-top fumigation chambers were constructed at Aberdeen University to a design specified by Ashenden et al. (1992). The chambers were constructed from clear corrugated PVC, with removable door panels and adjustable polycarbonate lids. Gas was distributed at ground level, through a perforated annular polythene tube. Chambers were arranged in pairs, each pair being served by a single pump via a split ducting pipe. The CO2 concentration was raised in one chamber of each pair by bleeding pure CO2 from a gas cylinder, via a needle valve and flow meter, into the ducting pipe downstream of the flow splitter. The other chamber of each pair received unaltered ambient air and therefore served as a control. In view of this construction, pairs of chambers are referred to here as experimental blocks (Mead et al., 1993).
CO2 concentrations in the chambers were measured using an ADC LCA-3 Infra-red gas analyser. The raised CO2 concentration was set at 600 ppm, the value predicted for the middle of the next century (Houghton et al., 1992), and monitoring studies of both short-term and diurnal fluctuations in this concentration were carried out. CO2 concentration was checked daily, and the flow rate adjusted if necessary. Temperatures in the chambers were monitored over two time periods, from 17 to 21 January 1994 and from 15 to 26 March 1994, using thermistor probes linked to a "Datahog" datalogger (Skye Instruments Ltd., Powys). Thermistors were sheltered from direct sunlight by tin-foil shields, and were placed amid the foliage of the Calluna plants in the chambers (see below). In order to minimise overheating inside the chambers due to greenhouse effects, the chamber lids were raised to their maximum height of 6 cm above the side-walls between 15 March and 1 October of each year. To conserve CO2, this height was reduced to 2.5 cm for the remainder of the year. Readjustment of the CO2 flow rate was carried out whenever lid height was changed.
-- Design of the experimental CO2 and nitrogen treatments
Eighty Calluna vulgaris plants, approximately 10 cm in diameter and aged approximately 5 years, were obtained from a moorland in NE Scotland (O.S. NO 644802, altitude 450 m) in February 1993. The plants were established individually in 18 cm diameter pots, using peat obtained from the same moorland site, and completed one growing season in an unheated glasshouse at ITE Banchory before being transported into the open-top chambers at Aberdeen on 1 November 1993. Twenty plants, with individual saucers, were placed in each of four chambers in two blocks, and CO2 fumigation of one chamber in each block commenced on this date. Ten plants within each chamber were randomly allocated to a raised nitrogen treatment, and the remaining ten served as controls.
The raised nitrogen treatment took the form of a 10 mM aqueous solution of ammonium nitrate which was applied weekly in seven equal doses starting on 17 March 1994. The amount of nitrogen applied to each plant on each treatment occasion was the equivalent of 0.5 g N m-2 , scaled to the surface area of the top of the pot, and the overall nitrogen treatment was therefore equivalent to 3.5 g N m-2. Control plants received equivalent volumes of water on each treatment occasion, and the positions of all pots within each chamber were re-randomised on these occasions. At other times, all plants were watered as often as necessary to prevent desiccation. The plants remained in the chambers for the whole of 1994; the length of current season's growth was measured weekly between 17 March and 19 June 1994 on two tagged shoots of each plant. The total number of flowers on each plant was determined on 13 July 1994.
In January 1995, the plants were re-potted into 27 cm diameter pots, again using moorland peat, in order to ensure continued potential for root growth. At this time, the experiment was expanded to include all eight chambers, in order to increase its statistical power to resolve CO2 main effects. The same eighty plants were used for this second phase of the experiment, with ten plants placed into each chamber. One chamber from each of the four blocks was allocated to raised CO2 treatment and one to ambient air, as before. Chamber allocations were re-randomised, with the constraints that all plants retained their original combination of CO2 and nitrogen treatments, and that there was still a balanced nesting of nitrogen treatments within chambers. Nitrogen treatments were applied starting on 7 April 1995. Seven equal weekly doses of ammonium nitrate solution were applied, giving a total dose of 7 g N m-2 .
-- Design of the O. brumata feeding trial
In the feeding trial, O. brumata larvae were allowed to feed on intact plants still growing in the fumigation chambers, rather than being reared in pots on excised foliage, since the latter technique can induce artefactual chemical changes in the plant tissue (Risch, 1985).
Adult O. brumata were captured at the Hunthill moorland outbreak (Fig. 2-1ii) during November 1993, and were stored at 5ºC in plastic jars, where they mated and oviposited on small rolls of filter paper. Broods of eggs were kept at constant 5ºC until 5 January 1994, when they were transferred to 1ºC to prevent premature hatching. A split brood design was employed in 1994: ten broods of forty eggs were obtained, each brood was split into eight batches of five eggs, and each of these batches was allocated to one plant from each of the eight block-treatment combinations. On 5 May 1994 each batch of five eggs was placed inside a 30 cm x 30 cm "Organza" polyester feeding bag, which was placed over approximately ten shoots of the designated Calluna plant and tied securely onto the woody stems at the base. Foliage was watered and allowed to dry prior to being enclosed in the feeding bags, in order to remove any accidental contamination from the ammonium nitrate applications. Care was taken to ensure that each feeding bag enclosed approximately the same amount of foliage, and that this amount was more than sufficient for the development of five larvae.
Larval emergence occurred from 22 May 1994 onwards, and larvae were seen to be adequately constrained by the feeding bags on this date. Twenty-eight days after this date, on 19 June 1994, feeding bags were removed and the enclosed foliage was searched for O. brumata larvae. Numbers of surviving larvae, their instar stage (Cuming, 1961) and fresh weight were recorded. Larvae were then put back into the feeding bags and replaced on the original plant, in order to permit further development to the pre-pupal stage. However, mortality in the feeding bags at the pre-pupal stage was too high to permit the recovery of pupal weight data in this year of the experiment.
The experimental technique was modified in 1995, since the allotted egg broods suffered a severe loss of viability, despite storage at 1ºC. Operophtera brumata larvae were therefore obtained from the Hunthill outbreak on 29 May 1995, and transported to the laboratory where they were classified into instar stages. Three early-third instar larvae were placed into each feeding bag and tied onto each plant. The four experimental blocks were set up progressively on four different days between 30 May and 4 June 1995 and initial larval size was standardised as closely as possible across treatments and blocks. Larvae were harvested sixteen days after the date of placement on each block, and larval weights were recorded. To facilitate development to pupation, larvae were placed into 7 cm diameter plastic jars which contained a 3 cm-deep layer of damp sieved peat. Larvae from each feeding bag were reared in separate jars. Fresh cut shoots from the appropriate C. vulgaris plant were supplied to each jar every 48 hours. Jars had porous lids, and were stored in shade at approximately 12ºC. Pupation was more successful under these conditions than in 1994, and the resultant pupae were sieved out of the peat eight weeks after harvest date and weighed.
-- Chemical analysis of Calluna foliage from the feeding trial
Samples of foliage were taken from each Calluna plant on 22 June 1994 for analysis of water content and total nitrogen and carbon concentrations. Shoots were excised at the base, and the current year's growth was separated from old foliage by dissection. Samples of both new and old growth were weighed, washed with deionised water and dried for three days at 80ºC. Samples were re-weighed, and water content of each sample was calculated by subtracting the ratio of dry to fresh weight from unity. Samples of new growth were then ground in a ball mill, and re-dried prior to chemical analysis. Total nitrogen and carbon concentrations were determined using a Fisons NA 1500 NCS autoanalyser, calibrated with a sulphanilamide standard. Vinyl gloves were worn for all stages of the protocol. Samples were randomised prior to allocation to different runs of this autoanalyser, in order to avoid systematic bias in the results due to calibration errors.
-- Analysis of data from the feeding trial
Generalised linear models were fitted to all measurements, using the GLIM (Royal Statistical Society, London, 1992) statistical modelling package. In order to avoid pseudoreplication (Hurlbert, 1984), CO2 main effects were tested for significance against an error term which described overall between-chamber variation, and which was obtained by pooling the block and block-CO2 interaction terms. Since the nitrogen treatment was nested within chambers, the significance of this main effect was tested against residual between-plant variation. Nitrogen-CO2 interaction and brood effects were also tested against this residual.
Larval growth rate was calculated from square root transformed larval weight data (see Appendix I). Larval survival in the 1994 experiment was expressed as counts out of the original five eggs for each feeding bag. A binomial error structure was specified in order to model this response (Crawley, 1993).
Larval development on the date of harvest, 19 June 1994, was described by a development index, as used previously (Chapters 3 & 5); this index was calculated for each feeding bag as follows:
Development index = ![]()
where i = instar stage
pi = proportion of larvae in feeding bag in instar i.
This response was also analysed using a binomial model.
Nine samples of Calluna shoots were taken on 6 June 1995 from the Queenamidda outbreak site on Orkney (O.S. HY 371210) (see Section 2.3.1), which was severely defoliated in 1980-1981 (Picozzi, 1981), but which was free from O. brumata in 1994 and 1995 (J.E. Kerslake, pers. obs.). On the same date, seven Calluna samples were taken from the Swartaback outbreak on Orkney (Fig. 2-1i), which still supported high densities of larvae (Fig. 3-11ii). Single samples were also taken on this date from two undamaged Orkney moorland areas, both at least 2 km from the nearest known O. brumata outbreak, at HY 318045 (altitude 80 m a.s.l.) and HY 386064 (altitude 40 m a.s.l.).
Samples were obtained by placing a 20 cm x 20 cm quadrat at random over an area of Calluna within the designated site; all Calluna shoots within each quadrat were cut and removed to a nearby laboratory, where the length of the current year's growth was measured. This current year's growth was then excised, washed, dried and ground as above (Section 7.2.1). Total nitrogen concentration was determined using a Fison's NA 1500 NCS autoanalyser, calibrated with a bulked soil standard.
Both the Orkney Calluna samples from the present investigation and the mainland Calluna samples used in Chapter 5 (see Section 5.2.4) were passed on the same date through the same run of the above autoanalyser. There was no reason to expect the mainland Calluna plants to be nitrogen-enriched, and the site from which they were taken was not subject to insect outbreaks during this study (Section 5.2.3). Consequently, the mainland Calluna samples provided a second set of control values against which to test for nitrogen enrichment of the Orkney moorlands.
In order to compare the Orkney and mainland data, it was necessary to standardise developmental stages of the two sets of plants (since Calluna tissue nitrogen concentration falls as the shoots extend: Fig. 5-7i). This was done by calculating the mean current year's shoot length of the Orkney outbreak samples; interpolation at this length onto a regression of nitrogen concentration against shoot length for the mainland plants (calculated from data in Chapter 5) gave a developmentally-matched tissue nitrogen value for mainland Calluna, against which to compare the Orkney data.
Raised CO2 concentrations in the open-top chambers fluctuated between 550 ppm and 700 ppm on a short-term scale (Fig. 7-1), and these fluctuations coincided with wind gusts outside the chambers (J.E. Kerslake, pers. obs.). There was no significant trend in the mean CO2 concentration in either chamber over this time scale (F(1,23) = 0.79 and 0.17, n.s.).
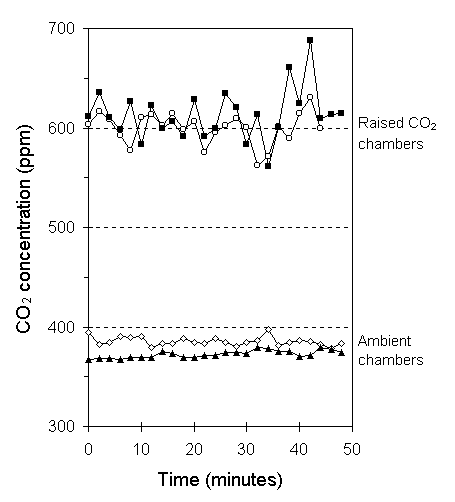
Figure 7-1. Short-term variation in CO2
concentrations inside four open-top chambers, two of which were subject
to experimental CO2 enrichment. Each series of points
represent a run of single readings taken at 2-minute intervals. Runs of
readings were taken at different times for each chamber, between 3 p.m.
and 8 p.m. on 19 March 1994.
Longer-term fluctuations in the mean chamber CO2 concentration were observed (Fig. 7-2), with the highest mean concentrations of approximately 630 ppm occurring around midnight, and the lowest of 530 ppm shortly after midday. This pattern of variability is consistent with the expected diurnal variation due to changes in photosynthesis rates (Leadley & Drake, 1993), although the magnitude of the variation was larger than that found in a similar study by Ashenden et al. (1992).
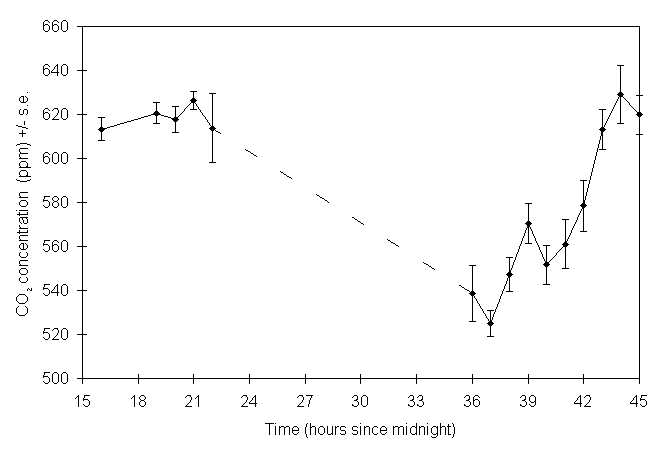
Figure 7-2. Variability in enriched CO2
concentration inside one open-top chamber in a 45-hour period from 20 to
21 March 1994. Points and bars represent the means and standard errors
of five readings taken at 2-minute intervals. The overall mean CO2
concentration over this period was 588.5 ppm (s.e. = 9.4 ppm).
Temperatures inside the chambers were frequently higher than those outside (Fig. 7-3), and Figure 7-4 suggests that two separate warming mechanisms operated at either extreme of the ambient temperature range. High ambient temperatures around midday were associated with a strong chamber warming of up to 8ºC (Fig 7-4). Greenhouse warming as a result of direct solar radiation was the most probable cause of this effect, which was larger than the 2 to 4ºC warming reported by Ashenden et al. (1992). A warming effect of up to 3ºC was also observed at low night-time temperatures (Fig. 7-4), although its cause was unclear.
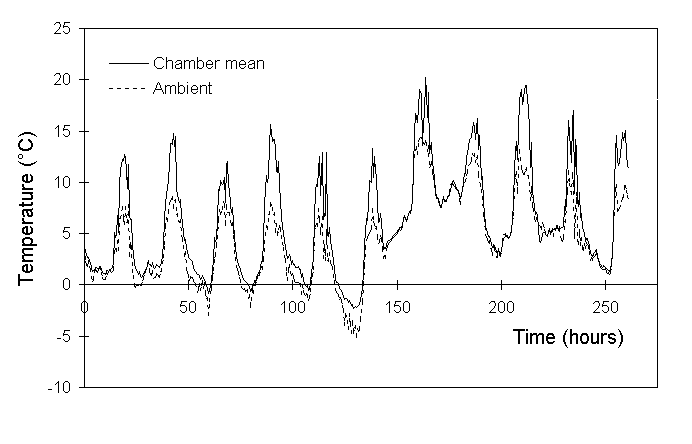
Figure 7-3. Mean temperature inside the open-top chambers (n
= 4) and ambient temperature adjacent to the chambers (n = 1) recorded
at 30-minute intervals between 15 and 26 March 1994.
Figure 7-4.
Mean chamber temperature (n = 4) plotted against ambient temperature (n
= 1) 15 to 26 March 1994. Solid line (y = x) represents equality between
the two temperatures.
There was also a small but consistent "edge effect" inside the chambers, with air temperatures at foliage level approximately 0.5ºC warmer for plants nearest to the chamber walls than for those at the centre (Fig. 7-5) over the 5ºC to 10ºC range.
Figure 7-5.
Temperature at the edge of a single growth chamber plotted against
temperature at the centre of the same chamber, 17 to 21 January, 1994.
Solid line represents equality between the two temperatures.
-- Shoot lengths and flowering
There was no significant main effect of raised CO2 concentration on the length of current year's shoots of Calluna on any date in 1994 (Fig. 7-6i). There was no significant main effect of nitrogen treatment on shoot length at the time of O. brumata hatching on 22 May 1994 (F(1,71) = 2.4, n.s.) (Fig. 7-6ii), but by the time of harvest on 19 June, a strong positive effect of nitrogen treatment was apparent (Fig. 7- 6ii).
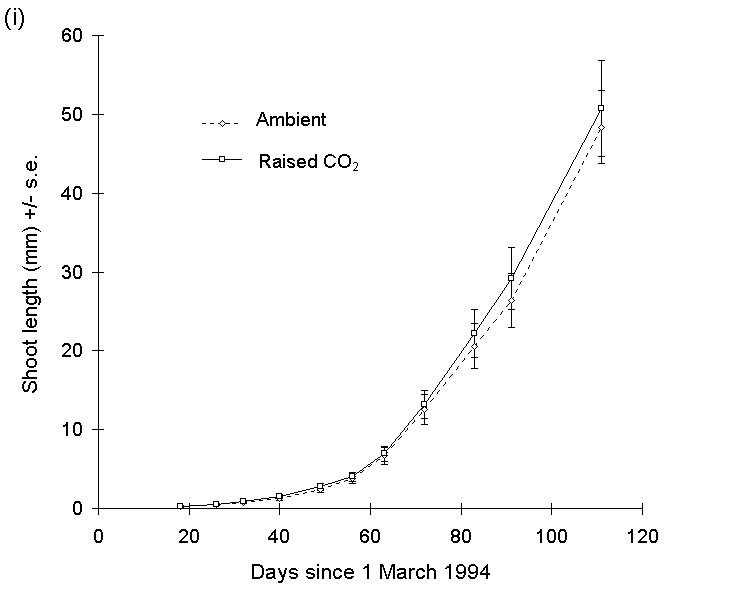
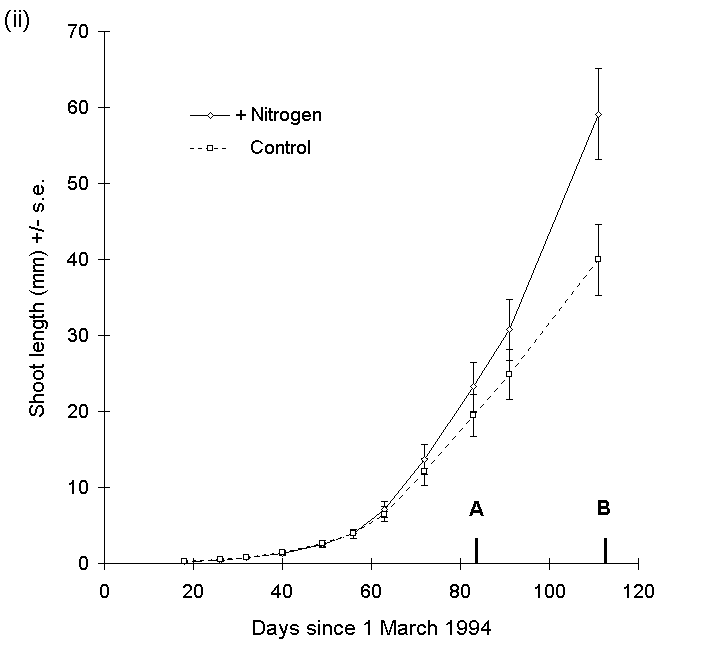
Figure 7-6. Mean shoot extension (+/- s.e.) of Calluna plants with time, 1994. (i): current year's shoot lengths for all raised-CO2 plants and all ambient CO2 plants. (ii): current year's shoot lengths for all nitrogen-enriched plants and all unfertilised control plants. Points A and B represent dates of emergence (22 May) and harvest (19 June) of O. brumata larvae respectively. Effect of nitrogen is significant at O. brumata harvest date: F(1,70) = 12.28, p < 0.001.
Covariance analysis of current year's shoot extension rate between O. brumata hatch and harvest dates, with shoot length at the former date as the linear covariate, showed a highly significant main effect of nitrogen treatment (F(1,69) = 12.74, p < 0.001). Nitrogen fertilisation, therefore, produced an increase in shoot extension rate once new shoots had begun to grow, but did not affect the date of inception of new growth (Fig. 7-6ii). There was no significant CO2 -nitrogen interaction on Calluna shoot length at harvest date (F(1,70) = 0.56, n.s.), and hence no evidence for nutrient-limitation of Calluna growth response to CO2 (see Section 1.6).
The above pattern was repeated in 1995, with no effect of CO2 on shoot extension, no CO2 -nitrogen interaction effect, no main effect of nitrogen on the date of first shoot production, but a significant positive main effect of nitrogen treatment on shoot length by 30 May (F(1,70) = 4.407, p < 0.05) (Fig. 7-7).
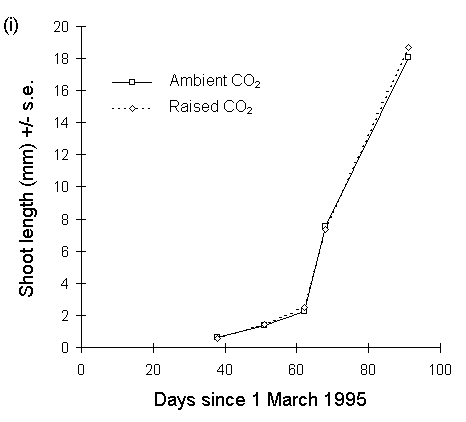
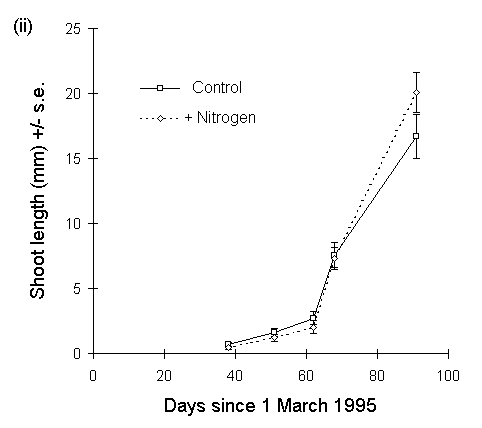
Figure 7-7. Mean shoot extension (+/- s.e.) of Calluna plants
with time, 1995. (i): current year's shoot lengths for all raised-CO2
plants and all ambient CO2 plants. (ii): current year's
shoot lengths for all nitrogen-enriched plants and all unfertilised control
plants. Effect of nitrogen is significant for final date (30 May): F(1,70)
= 4.41, p < 0.05.
There was no significant effect of either raised CO2 or nitrogen treatments on the number of flowers produced by the Calluna plants (Fig. 7-8).
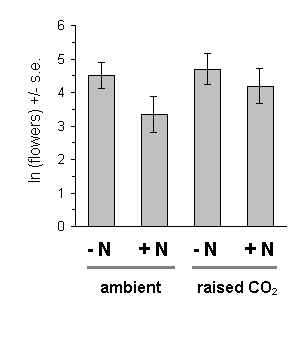
Figure 7-8. Flowering of C. vulgaris under raised or ambient
carbon dioxide concentrations, with raised nitrogen (+N) and control (-N)
treatments. Bars show means of logarithm of total number of flowers per
plant on 13 July 1994 (+/- s.e.). Effect of raised CO2: F(1,2) = 0.788,
n.s.; effect of raised nitrogen: F(1,74) = 3.022, n.s. (Note that standard
errors in this and subsequent figures are taken directly from the data,
not from the pooled variance of the generalised linear model).
-- Tissue chemistry
There was no significant effect of the raised CO2 treatment on Calluna C:N ratio (Fig. 7-9). However, nitrogen fertilisation caused a highly significant reduction in the C:N ratio of Calluna current year's growth (Fig. 7-9). Mean absolute total nitrogen concentrations were 2.81% (s.e. 0.168 %) in nitrogen-treated plants and 1.77 % (s.e. 0.096 %) in untreated plants.
Nitrogen treatment also resulted in significantly increased water content of both current year's shoots and previous years' foliage (Fig. 7-10). The physiological cause of this effect is unclear, although it has been observed in previous studies of Calluna nitrogen fertilisation (V.K. Brown. pers. comm.). There was no effect of CO2 on foliar water content.
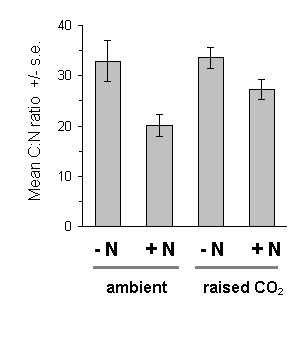
Figure 7-9. Carbon:Nitrogen ratio of C. vulgaris current year's
foliage on 22 June 1994. Main effect of CO2: F(1,2)
= 0.214, n.s.; nitrogen: F(1,72) = 34.51, p < 0.001;
CO2 -nitrogen interaction: F(1,72)
= 0.40, n.s..
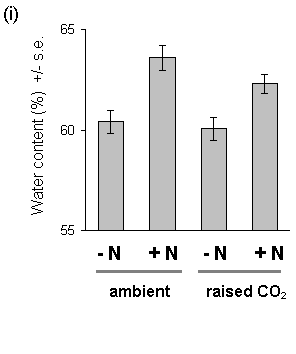
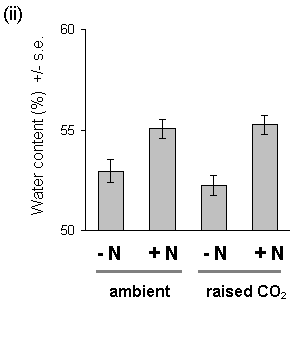
Figure 7-10. Water content of C. vulgaris foliage, 1994. (i):
current year's shoots; main effect of CO2: F(1,2)
= 5.67, n.s.; nitrogen: F(1,74) = 22.53, p < 0.001.
(ii): old foliage; CO2: F(1,2)
= 0.196, n.s.; nitrogen: F(1,74) = 20.39, p < 0.001.
-- Larval survival
The raised CO2 treatment had no significant effect on O. brumata survival to harvest in 1994 (Fig. 7-11). There was no significant effect of the nitrogen treatment on survival (Fig. 7-11), and no direct relationship between survival and the measured C:N ratio in the harvested foliage (F(1,63) = 1.44, n.s.). There was a significant brood effect on survival in 1994 (F(9,67) = 2.28, p < 0.05), but because "survival" in this case was uncorrected for initial hatching numbers, this effect could have been due to inter-brood variation in egg viability.
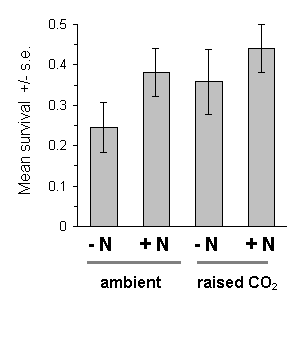
Figure 7-11. Larval survival on C. vulgaris at harvest date,
19 June 1994. Logit link analysis: CO2: F(1,67)
= 1.71, n.s.; nitrogen: F(1,67) = 2.59, n.s.; brood:
F(9,67) = 2.28, p < 0.05
There was a highly significant positive relationship between O. brumata
survival and C. vulgaris current year's shoot length at harvest
(logit link analysis, F(1,74) = 10.61, p < 0.01)
(Fig. 7-12). Some caution is necessary in the interpretation of this result,
since shoot extension was itself stimulated by nitrogen application (Fig.
7-6ii). However, a similar significant effect was observed (F(1,63)
= 11.1 p < 0.01) when shoot length at the time of hatching was fitted
as the final term of a logit survival model which already included the
measured C. vulgaris tissue C:N ratio, as well as CO2
and brood effects (Table 7-1). Shoot length then accounted for 15% of the
residual deviance. These results imply that there was a weak but significant
positive effect of Calluna current year's shoot length on O.
brumata survival, operating independently of plant tissue nitrogen
concentration.
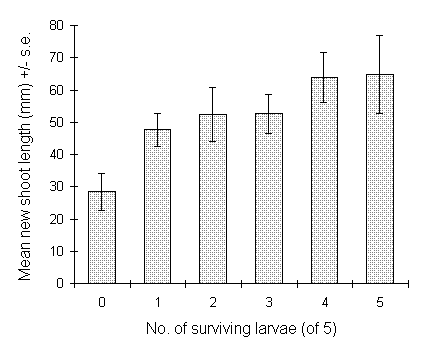
Figure 7-12. Lengths of current year's shoots of C. vulgaris
associated with each class of larval survival at date of harvest, 19 June
1994.
Table 7-1. Analysis of deviance table for larval survival, 1994, showing significant effects of brood and C. vulgaris current year's shoot length at time of hatching. (� CO2 main effect is tested here against the between-plant residual, since the between-chamber residual is anomalously low. This is the more rigorous test).
| Factor | d.f. | deviance | mean deviance | F Ratio | p |
| CO2 treatment |
1 |
4.318 |
4.318 |
2.340 � |
n.s. |
| Block + CO2 . block (resid. between chamber) |
2 |
0.183 |
0.091 |
||
| Foliar C:N ratio |
1 |
2.665 |
2.665 |
1.444 |
n.s. |
| Foliar C:N ratio . CO2 |
1 |
3.125 |
3.125 |
1.694 |
n.s. |
| Brood |
9 |
38.66 |
4.296 |
2.328 |
< 0.05 |
| Shoot length at time of hatch |
1 |
20.48 |
20.48 |
11.10 |
< 0.01 |
| Resid. between plants |
63 |
116.26 |
1.845 |
||
| Total |
78 |
185.69 |
To determine the developmental stage at which the above effect was most likely to have operated, the predictive power of shoot length on ultimate larval survival was determined by logistic regression, using data sets from each of the eleven shoot length measurements taken during the 1994 growth season, the last three of which spanned the duration of the feeding experiment. Shoot length at each date was a significant predictor of ultimate larval survival (Fig. 7-13).
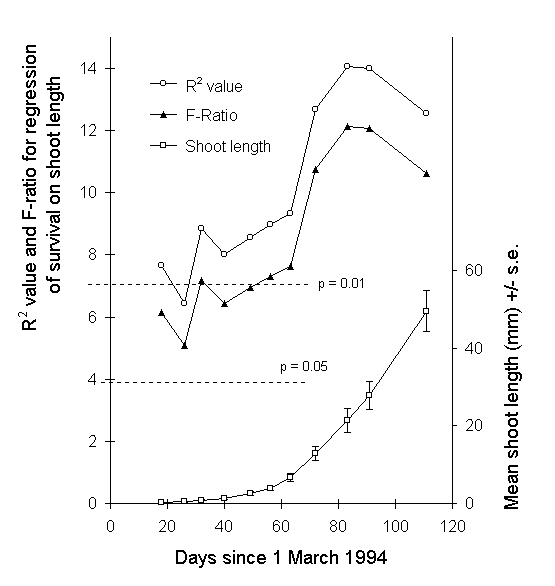
Figure 7-13. Upper lines: R2 values (%) and F-ratios
for the logistic regression of larval survival against C. vulgaris shoot
length, using shoot length data from each date shown. Upper and lower dotted
horizontal lines indicate 1% and 5% significance thresholds for the F-ratio
respectively. Lower curve: C. vulgaris mean shoot length at each time point,
with all treatments pooled.
The strongest predictive effect of shoot length on survival was obtained from shoot length measurements taken on 22 May 1994 (day 83 in Fig. 7-13), the same date on which O. brumata hatching was first observed in the feeding bags. This observation suggests that the effect of shoot length on survival operated most strongly at this early stage of larval life. This conclusion is supported by the fact that survival in 1995, in which year larvae were introduced to the feeding trial at the third instar, was uniformly high (c. 90%).
-- Larval growth rate, development and pupal weight
There was strong evidence for a large main effect of Calluna nitrogen enrichment on O. brumata development. The nitrogen treatment produced highly significant increases in larval growth rates both in 1994 and in 1995 (Fig. 7-14), a highly significant advancement of larval development (Fig. 7-15) and increased pupal weight (Fig. 7-16).
Growth of Calluna under raised CO2 conditions produced no significant main effect on any aspect of O. brumata development, either in 1994 or in 1995. There was a significant CO2 -nitrogen interaction effect on pupal weight in 1995 (Fig. 7-16), but the size and significance of this effect were marginal. Since no other significant CO2 -nitrogen interaction was found in this study, it is unlikely that the above effect on pupal weight is of physiological importance.
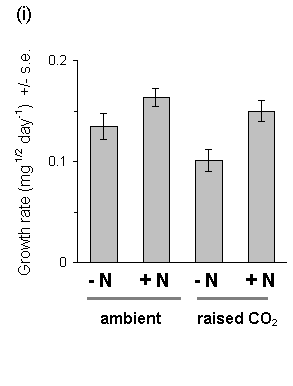
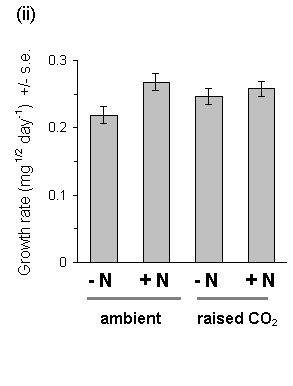
Figure 7-14. O. brumata larval growth rate on C. vulgaris in
(i) 1994 (egg to 4th instar) and (ii) 1995 (3rd to 5th instar). Growth
Rate was calculated as: [(final weight)1/2
- (initial weight)1/2]/ feeding time in
days*. Significance of main effects: 1994: CO2: F(1,2)
= 2.73, n.s.; nitrogen: F(1,54) = 10.7, p < 0.01;
1995: CO2: F(1,6) = 0.25, n.s.;
nitrogen: F(1,70) = 7.65, p < 0.01 ( * see Appendix
I).
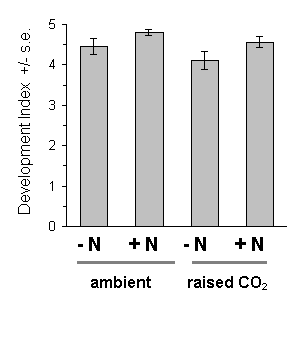
Figure 7-15. Development index of O. brumata on C. vulgaris after
28 days feeding. Effect of raised CO2: F(1,2)
= 2.14, n.s.; nitrogen: F(1,45) = 131.6, p < 0.001;
brood: F(9,45) = 1.91, n.s.
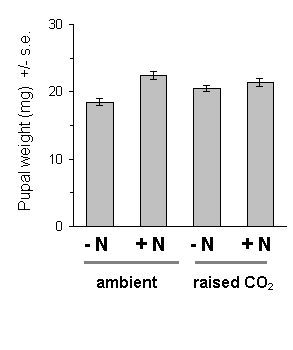
Figure 7-16. Pupal weights of O. brumata on C. vulgaris, 1995.
Effect of raised CO2: F(1,6)
= 0.006, n.s.; nitrogen: F(1,69) = 10.21, p < 0.01;
CO2 -nitrogen interaction: F(1,69)
= 4.09, p < 0.05
Males achieved slightly higher mean pupal weights than females (22.0 mg for males, 19.5 mg for females; F(1,69) = 9.21, p < 0.01), but there was no difference between sexes in the response of pupal weight to nitrogen treatment (nitrogen-sex interaction: F(1,69) = 0.55, n.s.). Pupal weights were low in comparison to those commonly attained by O. brumata on broadleaved hosts (Feeny, 1970; Wint, 1979).
Mean Calluna total nitrogen concentration in the current season's shoots was slightly above 2% dry weight at both of the Orkney outbreak sites, and similar values were obtained from the two control Calluna samples from non-outbreak sites on Orkney (Fig. 7-17).
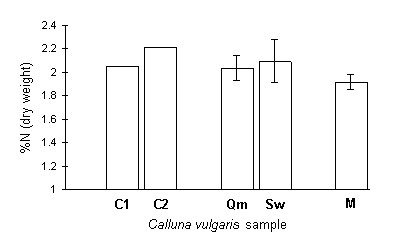
Figure 7-17. Total nitrogen concentration (% dry weight)
of C. vulgaris samples from five locations:
C1 & C2 : non-outbreak control sites on mainland Orkney, 6 June
1995;
Qm : Queenamidda, old outbreak site, Orkney, 6 June 1995 (+/- 95% c.i.);
Sw : Swartaback current outbreak site, Orkney, 6 June 1995 (+/- 95%
c.i.);
M : Mainland non-outbreak site C. vulgaris plants (+/- 95% c.i.)
(calculated as described in Section 7.2.2).
Calculation of a developmentally-matched value for the mainland Calluna plants (see Section 7.2.2) was performed as follows: mean current year's shoot length of the Orkney samples on 6 June 1995 was 7.88 mm (s.e. = 0.36 mm); regression of mainland Calluna nitrogen content against current year's shoot length (from data obtained in Chapter 5) gave the equation: %N = 1.95 - 0.0044 (shoot length) ; (R2 = 13%, p < 0.01). Interpolation at 7.88 mm gave the mainland value of 1.915% (s.e. = 0.032%) (Fig. 7-17 M), which did not differ significantly from the means of either of the Orkney outbreak samples (t74 = 1.31 for Qm; t72 = 1.67 for Sw; n.s.).
Manipulative feeding trials inevitably involve the exposure of plants and insects to artificial growth conditions. In this study, considerable warming of the CO2 fumigation chambers occurred (Fig. 7-4); this was unavoidable, since to have removed the chamber lids completely would have destabilized CO2 concentrations, and the use of shade netting would have reduced the photosynthesis rates of the plants. The extent to which O. brumata performance was affected by the increased chamber temperatures is difficult to assess, although the almost complete failure of larvae to reach a viable pre-pupal stage in 1994 suggests that chamber conditions were not ideal.
Despite the above limitation, the larval survival rates obtained in this study were similar to those obtained in another feeding trial conducted simultaneously on Calluna in a neighbouring outdoor plot (results presented in Chapter 5). This suggests that the chamber conditions were not sufficiently harsh to increase larval mortality over the feeding period.
Although raised CO2 concentrations showed considerable fluctuations with time (Fig. 7-2), this fluctuation was systematic and comparable in magnitude with the diurnal variations observed by Leadley & Drake (1993). Regular monitoring and adjustment of CO2 input flow rates in this study prevented any longer-term variation.
The failure of this study to demonstrate any significant effect of CO2 enrichment on Calluna growth or tissue chemistry agrees with similar work by Coop (pers. comm.), but disagrees with Woodin et al.(1992), who found significant stimulation of shoot growth and flowering under raised CO2 , together with a reduction in foliar nitrogen concentration. There are several possible reasons for this discrepancy in results. Firstly, the current study employed a more stringent experimental design and statistical analysis, in which chambers rather than individual plants within chambers were treated as replicates; such fully-replicated designs are not always possible for practical reasons, but substantially increase confidence that any observed CO2 effects are genuine (Hurlbert, 1984). Secondly, this study used wild plant material, taken directly from a high-altitude moorland site, as opposed to the horticultural cultivars studied by Woodin et al. (1992); the latter plants were fertilised and possibly selected for increased growth rates. In contrast, C. vulgaris in its wild state is a slow-growing, "risk-averse" perennial adapted to harsh, low-nutrient habitats (Aerts & van der Peijl, 1993), and consequently unlikely to respond rapidly to any form of enrichment (Chapin & Shaver, 1985; Loehle, 1995). Thirdly, distinct individual plants were used in this study, in contrast to the use of clonal cuttings by Woodin et al. (1992); use of clonal cuttings removes a source of variability from the experiment, but reduces the generality of the results.
Calluna was equally unresponsive to CO2 enrichment under both low and high-nitrogen treatments, since there were no significant CO2 -nitrogen interaction effects on any plant response. There was, therefore, no evidence in this study that the response of Calluna growth to CO2 enrichment was nutrient-limited (Woodin et al., 1992) (see Section 1.6).
In view of the evident unresponsiveness of Calluna to CO2 enrichment, the failure to demonstrate any significant CO2 main effect on O. brumata development on this host is not surprising. The present null result is consistent with the lack of significant CO2 effects on O. brumata herbivory on sycamore and beech (Watt et al., 1996), and indicates that the O. brumata-C. vulgaris system is unlikely to respond strongly or rapidly to future changes in CO2 concentrations. The implications of this conclusion for future outbreak potential are discussed in Section 7.5.
This study confirms the assumption made in Chapter 6, that fertilisation of Calluna with ammonium nitrate solution causes an increase in tissue nitrogen concentration, and provides a further demonstration that O. brumata development is positively affected by this treatment. Larval development and pupal weight were both increased on nitrogen-treated Calluna plants (Figs 7-15 & 7-16), as was larval growth rate (Fig. 7-14) (Note that the latter result disagrees with the stabilisation of growth rate shown in Chapter 6; although the reasons for this disagreement are unclear, it illustrates the difficulty of comparing larval responses between different experiments conducted under different conditions).
The positive response of O. brumata to Calluna nitrogen-enrichment is consistent with the hypothesis that moorland O. brumata outbreaks are caused by nitrogen enrichment in the field (Sections 1.2.3 & 7.1.3). However, several caveats apply to the interpretation of these results; the use of a chemical treatment to increase Calluna nitrogen concentration constitutes a severe manipulation, and could have induced unnatural changes in plant physiology. It was not within the scope of the present study to determine the chemical form in which the additional nitrogen was present in the plant tissue, although the increased water content of nitrogen-treated plants (Fig. 7-10) attests to a pronounced physiological change. It is possible that a proportion of the additional nitrogen was stored in a non-structural, soluble form in the foliage at the time of larval feeding. Such a pool of readily-accessible nitrogen could have been the primary reason for the observed stimulation of larval development, and may be rarely experienced by O. brumata in natural systems. Furthermore, the increased water content of the nitrogen-treated plants may have affected other aspects of diet quality, such as leaf toughness (Feeny, 1970). Direct effects of nitrogen enrichment are therefore potentially confounded with other unknown physiological changes in the foliage. These caveats reinforce the need to draw a distinction between responses to artificial manipulations of host quality (as in this study), and responses to natural variations in unmanipulated plants (as in Chapter 5).
This study demonstrated a significant predictive effect of Calluna current year's shoot length on ultimate larval survival, which acted independently of Calluna C:N ratio (Table 7-1). The relationship was significant even with data from the earliest measurement of shoot length in 1994, which was performed two months before the date of egg hatch, one day before the first application of the nitrogen treatment, and one month before the observation of any effect of nitrogen treatment on shoot growth rate (Fig. 7-13). A concerted positive effect of nitrogen-enrichment on both shoot length and survival was, therefore, not the cause of this relationship.
Individual variability in the date of inception of shoot growth is the most probable explanation for the above relationship; the availability of sufficient quantities of new season's shoots at the time of hatching may be a key requirement for the survival of O. brumata on C. vulgaris, with earlier-flushing plants better able to support the establishment of newly-emerged larvae (see also Section 3.4.1). The strongest predictive effect of shoot length on survival was obtained from data recorded on the date of larval emergence (Fig. 7-13), which supports this hypothesis.
The above data are interesting in the context of previous results: Section 3.3.1 showed that O. brumata larvae were not present on Calluna before the onset of new shoot growth, probably because the previous year's foliage is too tough or low in nutritional quality to permit larval establishment; Chapter 5 showed that larval survival on Calluna was not reduced by a hatch delay of one month. The latter result implies that there is no penalty for late hatching of O. brumata on Calluna, whilst the results of the present study suggest that there is a penalty for premature hatching. This is a logical conclusion, since premature hatching will result in larvae being unable to locate any current year's growth of C. vulgaris, whilst late hatching larvae will experience foliage that declines in quality more slowly than that of many broadleaved trees (Feeny, 1970; Wint, 1979) (Fig. 5-7i).
There was no evidence that Calluna from the Orkney outbreak sites had significantly higher tissue nitrogen concentrations than the control samples from the unaffected Orkney moors or the mainland plants (Fig. 7-17). Consequently, these results fail to support the hypothesis that O. brumata outbreaks are triggered by host nitrogen enrichment. However, the limited levels of replication in this study, and the need to standardise developmental stages between samples, argue that a more intensive sampling programme would be necessary in order to test this hypothesis conclusively.
The measurements of Calluna nitrogen concentration in the field reinforce the point made in Section 7.4.3, that the nitrogen fertiliser treatment in the feeding trial was a severe manipulation; fertilised Calluna plants in the feeding trial showed a mean nitrogen concentration of 2.81% , which was considerably greater than the highest value obtained in field-sampled vegetation of 2.21% (Orkney C2).
Field-sampled Calluna nitrogen concentrations were, nevertheless, high in comparison to many previously-published studies of Calluna in Scotland (Powell & Malcolm, 1974; Pitcairn et al., 1995; Iason et al., 1993; Hartley & Gardner, 1995), most of which give values of between 1.0 and 1.9% dry weight. This discrepancy is partly due to differences in assay technique: the studies cited above all employed "wet" assay techniques, in which the nitrogen content of samples was extracted by digestion in concentrated acid (see Cresser & Parsons, 1979). The present study, in contrast, used a NCS autoanalyser which combusted samples in their "dry" state, and this technique gives consistently higher values than acid digestion (see Appendix II). Using the relationship between these two assay techniques given in Appendix II, the Orkney Calluna data in Figure 7-17 can be converted into their "acid digest equivalent" values, which are 1.83% (s.e. = 0.048%) for Queenamidda, and 1.90% (s.e. = 0.080%) for Swartaback. These adjusted values are in agreement with the results of Powell & Malcolm (1974) for mature Scottish Calluna sampled in June. The lower values reported by other studies were obtained from Calluna sampled either in August (Iason et al., 1993; Hartley & Gardner, 1995) or during winter (Pitcairn et al., 1995), at which times foliar nitrogen content would be expected to be lower than in June (Brunsting & Heil, 1985).
In conclusion, the results of this study provide no evidence that Calluna from the O. brumata outbreak sites on Orkney has significantly higher total nitrogen concentrations than would be expected from previously-published studies, or by comparison with Calluna from control non-outbreak sites on Orkney and mainland Scotland.
Extrapolation of the results of manipulative experiments to the broader question of outbreak potential under future environmental change is a complex task. Insect outbreaks are essentially tri-trophic systems, since natural enemies have a profound influence on herbivore population dynamics (Berryman et al., 1987; Roland & Embree, 1995), yet few investigations have so far considered the impact of climatic change on population regulation by natural enemies (Landsberg & Smith, 1992; Hassell et al., 1993). In addition, environmental change can affect herbivore performance via plant growth and nutrient content (Lincoln et al., 1986), allocation to secondary defence compounds (Lindroth et al., 1993) and altered phenology (Uvarov, 1931; Dewar & Watt, 1992; Murray et al., 1994). Plant growth responses will themselves vary with nutrient availability (Bazzaz, 1990; Woodin et al., 1992). No single study can encompass the whole range of potential effects and interactions, and most manipulative experiments necessarily involve the sudden exposure of "today's" populations of plants and insects to "tomorrow's" environmental conditions, without allowing for the adaptive role of selection acting over successive generations. Since insect populations commonly display heritable adaptation to local plant phenology (Mitter et al., 1979; Du Merle, 1983; Komatsu & Akimoto, 1995), then "tracking" responses of insect populations to the effects of climate change on their hosts are likely to occur over many generations. If such tracking responses do occur, then current manipulative studies may simply serve to highlight the magnitude and direction of the tracking that will be required. Ultimately, the effects of climate change on weather variability (and hence predictability of plant phenology) could prove to be a better determinant of outbreak potential than the general responses of plant and insect development to simulated future climates (Thomson et al., 1984; Auerbach, 1991) (see Chapter 8 for further discussion).
In an attempt to resolve some of the above complexity, Landsberg & Smith (1992) developed a "functional scheme" to infer the susceptibility of natural systems to insect outbreaks under climate change regimes, based on the tri-trophic attributes of these systems. Their conclusions agree well with the results of the present study, and others (Watt et al., 1996), that increasing CO2 is unlikely to have a strong effect on the probability of outbreak of a generalist univoltine leaf-chewing insect such as O. brumata, particularly on a slow-growing host, rich in carbon-based secondary defence compounds, such as Calluna. Even if it is assumed that future long-term exposure of Calluna to rising CO2 levels will eventually produce a small reduction in tissue nitrogen concentration (Bazzaz, 1990), the remarkable ability of O. brumata to exploit this poor-quality host is likely to confer resilience to any such gradual decline in its nutritive quality (see Chapter 5).
Other results discussed above (Section 7.4.4) indicate that there is a penalty for early hatching of O. brumata on C. vulgaris, since larvae need to establish on new season's shoots. This suggests that the probability of outbreak would be reduced if the date of O. brumata hatching were to be advanced relative to the onset of Calluna shoot growth. Such an effect might be caused by differing responses of plant and insect phenology to a rise in ambient temperatures (Dewar & Watt, 1992). However, as discussed above, selection on hatching dates would operate strongly against such a discrepancy.
The positive effect of Calluna nitrogen fertilisation on O. brumata development and pupal weight in this study suggests that any future nitrogen enrichment of heathland vegetation will have the potential to trigger O. brumata outbreaks. Rapid larval development on nitrogen-enriched Calluna could reduce vulnerability to parasitoids (Cheng, 1970; Loader & Damman, 1991) or avian predators (Picozzi, 1981), whilst increased female pupal weight would result in increased potential egg production (Roland & Myers, 1987). Furthermore, stimulated Calluna shoot growth in response to nitrogen enrichment (Fig. 7-6ii) would increase the total quantity of food available to O. brumata. However, as discussed above (Section 7.4.5), there is no evidence that this mechanism is already responsible for the current moorland O. brumata outbreaks (see Section 7.4.5).
Exposure of Calluna plants to raised CO2 concentrations over two growing seasons had no significant effect on their growth and nutritional quality for O. brumata. There is, therefore, no evidence from this study that increasing atmospheric CO2 concentrations will greatly affect this system.
In contrast, nitrogen fertilisation of Calluna stimulated shoot growth rate and produced strong positive effects on O. brumata development. However, this fertilisation treatment increased Calluna nitrogen concentration beyond its natural range in the field, and may have induced unnatural changes in plant physiology.
The hypothesis that O. brumata outbreaks are caused by nitrogen-enrichment of moorland vegetation was not supported by assays of Calluna tissue nitrogen concentration in Orkney outbreak sites.
Operophtera brumata survival was positively related to Calluna current year's shoot length; availability of this new growth at the time of larval emergence may be an important requirement for O. brumata establishment on Calluna.
|
Previous Chapter |
 Contents Page |
Next Chapter |