|
Previous Chapter |
 Contents Page |
|
Previous Chapter |
 Contents Page |
The results of the previous chapters are reviewed in order to highlight the relative contributions of "bottom-up", "top-down" and climatic effects to the development of moorland O. brumata outbreaks. It is argued that the interaction between climate and the "top-down" effect of natural enemies is more important for determining outbreak potential than the "bottom-up" effect of host quality. A schematic model is presented to illustrate this argument. The effects of future climatic changes on the O. brumata- C. vulgaris system are considered, and shown to be complex and difficult to predict. It is argued that future changes in snow cover patters and natural enemy distribution will have particularly powerful effects on future outbreak potential.
Operophtera brumata has been present on British moorlands at sub-outbreak densities, feeding on Calluna vulgaris, for many years (Traill, 1869). Moorland O. brumata outbreaks are therefore not the result of a recent "host shift" (Fraser & Lawton, 1995), such as that which enabled O. brumata to colonise Picea sitchensis in Scotland (Fraser, 1995). In contrast, moorland O. brumata outbreaks in Scotland can be viewed as localised "releases" of previously-existing moorland populations, occurring at widely-spaced locations within extensive stands of heathland vegetation. The term "release" implies the lifting of a previous constraint: as discussed previously, such constraints include the "bottom-up" effect of low host-plant quality (Section 1.2), the "top-down" effects of natural enemies (Section 1.3.3), and direct effects of climate (Section 1.3.2). These effects are not necessarily mutually exclusive: release from more than one of the above constraints may be necessary for the occurrence of moorland O. brumata outbreaks, and different interactions may be important at different sites.
"Top-down" and "bottom-up" constraints were discussed by Hunter & Price (1992), who argued that any dichotomy between these two theories of herbivore population regulation is false: forces in both directions can operate simultaneously (see also Karr et al., 1992; Power, 1992). According to Hunter & Price (1992), top-down effects are superimposed onto a basic bottom-up trophic framework, with plant quantity and quality setting the overall carrying capacity for the higher trophic levels. "Abiotic heterogeneity" (e.g. climatic variability) can then influence the relative strength of top-down and bottom-up forces at each trophic level. This model, therefore, demands a synthesis of older top-down (Hairston et al., 1960) and bottom-up (Feeny, 1976; Rhoades, 1983; White, 1984) mechanistic views of herbivore population regulation, taking into account the effects of local environmental conditions at each individual study site.
With the above model in mind, the following sections review the conclusions of Chapters 3 to 7, and critically evaluate the evidence for bottom-up, top-down and climatic constraints on moorland O. brumata populations.
Calluna vulgaris is clearly a sub-optimal host for O. brumata, compared with the broadleaved shrub Vaccinium myrtillus (Chapter 3). The low nutritional quality and low digestibility of Calluna result in reduced larval survival and development. However, the occurrence of O. brumata outbreaks on stands of pure Calluna on Orkney demonstrates that the ability of O. brumata to attain high population densities, in contrast to its individual larval performance, is not greatly constrained by the low nutritional quality of Calluna.
This study found no evidence that the Calluna stands which have been subject to O. brumata outbreaks on Orkney possess unusually high tissue nitrogen concentrations (Section 7.4.5). However, nitrogen levels in Calluna tissue during the growing season are a "moving target" (Fig. 5-7i), and there are difficulties in knowing precisely when to sample the affected vegetation, and in obtaining suitable developmentally-matched control samples. Further sampling will certainly be necessary in order to clarify this issue. Nevertheless, the hypothesis that moorland O. brumata outbreaks are triggered by an increase in atmospheric nitrogen deposition, as proposed for Lochmaea suturalis outbreaks in Holland (Heil & Diemont, 1983), is difficult to reconcile with the geographical restriction of the recent O. brumata outbreaks to some of the least polluted areas in the U.K. (Pitcairn et al., 1995). Furthermore, the low pupal weights and small adult sizes of O. brumata reared on Calluna taken directly from the Hunthill outbreak (Chapter 3) argue against the presence of substantial nutrient enrichment of vegetation at this site.
Despite the low nutritional quality of Calluna, Chapter 5 demonstrates that O. brumata survival and development are not adversely affected by a further decline in this nutritional quality. Operophtera brumata is clearly capable of completing its development successfully on host foliage that is lower in nitrogen concentration than the young Calluna foliage that it normally encounters in the field (Figs 5-7i & 7-17). Furthermore, the demonstration that O. brumata is capable of compensatory feeding (Chapter 6) supports the view that O. brumata is a versatile herbivore, and is able to buffer its development against fluctuations in host quality (Section 5.4.2). In this respect, O. brumata conforms well to the description of a highly adaptable generalist herbivore given by Price (1992) (see Section 1.3.1), a view which is reinforced by the failure of the present study to demonstrate any nutritional specialisation of moorland and oak woodland populations to their respective hosts (Section 4.4.3) (see also Ruohomäki & Haukioja, 1992b).
In contrast, experimental nitrogen fertilisation of Calluna plants did elicit a significant positive response in O. brumata development on two occasions (Chapters 6 & 7), and this was associated with a significant increase in Calluna foliar nitrogen concentration on at least one of these occasions (Section 7.3.2). This appears to contradict the above conclusion that O. brumata is resilient to changes in host quality. However, it is necessary to consider both the magnitude and the direction of the changes involved: in Chapter 5, O. brumata showed no negative response to a natural decline of 15% in tissue nitrogen concentration, whilst in Chapter 7, O. brumata showed a positive response to an artificially-induced 60% increase in tissue nitrogen concentration. These apparently contradictory responses could be explained by the model presented in Section 5.4.2: nutritional compensation, operating strongly at low host quality, allows O. brumata to buffer its development against a further small reduction in host quality, whilst still retaining the ability to respond positively to a more substantial increase in quality. Both the magnitude and the direction of changes in host quality may therefore be important in determining the herbivore response (Fig. 5-9).
Alternatively, the differing responses above can be explained as artefacts of methodology: application of aqueous ammonium nitrate is a severe manipulation, and could have produced unnatural physiological changes in the plants (Section 7.4.3). Artificially increasing Calluna tissue nitrogen concentration by 60% could have created a pool of soluble, non-structural nitrogen stored in cell vacuoles (S. Woodin, pers. comm.), which would rarely occur in Calluna in the field.
The observation that O. brumata development is improved by host nitrogen enrichment is not surprising: as Hanhimäki et al. (1995) point out, "there is rarely compensation for high-quality diet". However, it does not automatically follow that host quality is currently the most important factor limiting O. brumata densities in the field. Experimental manipulation of Calluna nutrient content in situ would be the most direct test of the latter hypothesis (see Hartley & Gardner, 1995), and is recommended in any future studies. In addition, other aspects of host quality besides foliar nutrient content should be considered: the rôle of plant secondary defence chemicals, which it has not been possible to assess in the present study, is clearly worthy of investigation (Feeny, 1968, 1976; Haukioja, 1980).
In summary, the majority of the available evidence suggests that "bottom-up" effects of moorland host-plant nitrogen concentration are unlikely to have a strong limiting effect on O. brumata outbreak potential in the field: O. brumata achieves larval densities of at least 600 m-2 on Vaccinium-poor moorland, and 1400 m-2 on Vaccinium-rich moorland (Fig. 3-11), with as yet no evidence that these sites are nutrient-enriched. If host quantity and quality determine a system's potential herbivore carrying capacity (Hunter & Price, 1992), then the instantaneous carrying capacity of unenriched moorland vegetation for O. brumata appears to be extremely high. However, this potential carrying capacity is only actually attained during localised, sporadic outbreaks (Price, 1992), which implies that other non-nutritional constraints must usually operate to keep moorland O. brumata densities low.
The striking difference between the parasitism levels in high-altitude and low-altitude moorland O. brumata populations demonstrated here (Section 4.3.5) provides evidence that the differing climates of these two moorland types modify the relative importance of this "top-down" effect. Escape from parasitism may be more common on high-altitude moorlands than on Orkney, since the harsh climatic conditions of high-altitude sites can inhibit parasitoid foraging (Coulson & Whittaker, 1978). A similar effect of altitude-related "abiotic heterogeneity" on parasitism levels of Phyllonorycter sp. (Lepidoptera: Gracillariidae) was found by Preszler & Boecklen (1996). Their conclusion, that the strengths of "top-down" and "bottom-up" trophic effects on herbivores vary under differing abiotic environmental conditions (as proposed by Hunter & Price, 1992), is therefore in agreement with the results of the present study.
The data presented in Figure 4-11 provide only a single year's assessment of parasitism rates. A longer-term study of parasitism in Orkney moorland O. brumata populations would be instructive, in order to determine whether outbreaks at these sites develop in the continual presence of substantial levels of parasitism. Alternatively, the outbreaks on Orkney may occur as a consequence of a temporary "release" of O. brumata populations from parasitism, which is followed in subsequent years by a progressive re-exertion of this "top-down" constraint. The latter hypothesis is strengthened by the observations of Lorimer (1988) that the level of parasitism in an O. brumata outbreak population on Orkney in 1981 was only 2%, but had risen to 60% by 1987 ‡ . Further monitoring of O. brumata population densities at the Swartaback outbreak site would be of interest, in order to determine whether the outbreak there will persist in the continued presence of relatively high parasitism levels.
Evidence is presented in Chapter 3 that climatic variability acts as a major constraint on high-altitude moorland O. brumata populations: disruption of adult emergence by early-lying snow was associated with a sharp decline in O. brumata abundance at the Hunthill site between 1993 and 1994 (Figs 3-10ii & 3-11i). This effect may exert a selection pressure in favour of earlier adult emergence at high altitudes (Schneider-Orelli, 1916), and the observation of this life history trait in a high-altitude moorland O. brumata population (Fig. 4-4) strengthens the view that early snowlie is an important limiting factor for this population.
The importance of climatic variability at the low-altitude Orkney sites is less easily assessed, in the absence of climatic data from these locations. However, the reported rarity of long-lasting snow cover at these sites during November (E. Meek, pers. comm.), combined with the later adult emergence of Orkney O. brumata populations (Fig. 4-4), suggests that this effect is of less significance on Orkney.
In view of the above conclusions, a schematic model which may help to explain the occurrence of high-altitude moorland O. brumata outbreaks is as follows:
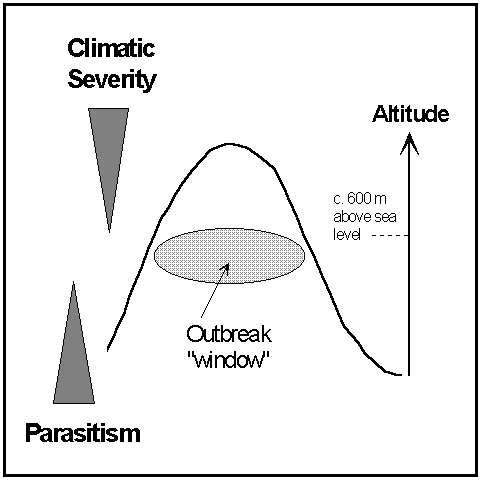
Figure 8-1. Schematic model to explain the occurrence of high-altitude
moorland O. brumata outbreaks. "Windows" may be created for O.
brumata populations between the constraints of climatic severity and parasitism.
Width of the arrows indicates the altitudinal variation of the above constraints.
It is proposed that moorland O. brumata populations are constrained
at high altitudes by climatic severity (in particular, the disruptive effect
of snow cover on adult emergence), and at lower altitudes by parasitism.
These constraints vary in strength with altitude, but in opposite ways
(Fig. 8-1). The relaxation of one or both of these constraints will therefore
reveal temporary "windows" of enemy-free space in which O.
brumata populations can establish. In such windows, the potential carrying
capacity for O. brumata will be set by the quality and quantity
of available vegetation, as described above (Section 8.1.2) (Hunter &
Price, 1992). If climatic conditions remain favourable for several successive
years, and if parasitoids do not extend their range upwards into the developing
O. brumata population (i.e. if the window stays "open"),
then an outbreak may occur. In some years (as in winter 1993 at Hunthill,
Fig. 3-10), unfavourable climatic conditions can effectively "shut"
all such windows, causing a sharp decline in high-altitude O. brumata
abundance.
Initial establishment of O. brumata populations in new "windows" will occur by reproduction of indigenous high-altitude individuals, previously present at low densities, or by wind-assisted immigration of young larvae from lower altitudes (possibly even from populations on other host species). Lower-altitude Vaccinium myrtillus patches could act as "refuges" in which residual O. brumata populations can persist, and from which they will colonise higher-altitude Calluna-rich vegetation in favourable years. Lower-altitude O. brumata populations may gain greater protection from parasitism on Vaccinium than on Calluna, since the broadleaved structure of the former plant enables larvae to spin the leaves around themselves more tightly.
Once a newly-opened "window" is colonised, selection during the course of population build-up will favour the establishment in the population of the high-altitude life history traits described in Chapter 4.
The above model does not include effects of predation, disease and host-plant chemical defences. Although studies of other insect-plant systems have highlighted the importance of these effects (Haukioja, 1980; Roland, 1990; Myers & Kuken, 1995), too little is currently known about their spatial and temporal patterns of operation in moorland O. brumata populations. Furthermore, the above model may not apply to outbreaks on low-altitude Orkney moorlands, where the rôles of climatic severity and escape from parasitism are less clear (see Sections 8.1.3 & 8.1.4). Nevertheless, the model may serve to stimulate further investigation of the relationships between climatic variability, natural enemy pressure and O. brumata abundance as a function of altitude (see Preszler & Boecklen, 1996).
Given that the causes of moorland O. brumata outbreaks are still unclear (Section 8.1), attempts to predict their future frequency and severity under conditions of environmental change are obviously difficult (see Section 7.4.6). The above discussion (Section 8.1) highlights the need to consider the effects of future environmental change not solely on the "bottom-up" interaction between host quality and insect performance (as in Chapter 6), but also on "top-down" natural enemy pressures, and on the variability of weather patterns. Some of these interactions may be more predictable than others, as discussed below.
In view of the number of interactions which need to be considered when predicting the future outbreak potential of insect herbivores, Landsberg & Smith (1992) stressed the need to focus on key "functional attributes" of the systems in question, including the role of natural enemies. A "functional scheme", based upon the examples given by Landsberg & Smith (1992), is presented here for the O. brumata-C. vulgaris interaction (Table 8-1). The potential effects of increased CO2 concentrations, increased nitrogen deposition and increased temperature are considered (Bazzaz, 1990; Vitousek, 1992), together with those of increased winter precipitation and reduced summer precipitation (as predicted for the U.K. by Department of the Environment, 1991). The final column of Table 8-1 summarises the most likely effect of this suite of environmental changes on the probability of outbreak.
It is immediately apparent from Table 8-1 that many of the effects under consideration cannot be predicted with any degree of certainty, due to lack of data. In other cases, different environmental changes may affect the system in opposing ways. However, some generalisations can be made, based upon the results of the present study:
In view of the lack of significant response of the O. brumata-C. vulgaris system to experimental CO2 enrichment (Chapter 7) or raised plant growth temperatures (Chapter 6), and also considering the lack of importance ascribed to "bottom-up" effects of host-plant quality in determining outbreak distribution (Section 8.1.2), it is proposed that the most powerful effects of environmental change will operate on the system by mechanisms other than host-plant quality. If future increases in nitrogen deposition to Scottish moorlands do have an effect on O. brumata abundance, it will probably be a positive one (by analogy with Heil & Diemont, 1983; Hartley & Gardner, 1995), but it is important to restate the point that O. brumata outbreaks have already been extremely damaging in some of the most apparently unpolluted sites in the U.K.
Disruption of the phenological relationship between O. brumata larval emergence and C. vulgaris budburst could be important if temperatures become more variable, since there is evidence that premature larval emergence results in increased mortality (Section 7.4.4). Year-to-year variability in temperatures could be more important than overall long-term temperature trends, since O. brumata hatching dates may evolve to "track" the latter effect (Holliday, 1985; Dewar & Watt, 1992). The availability of Vaccinium myrtillus will, however, greatly reduce the penalty for early hatching (Fig. 3-4), and will therefore counteract the above effect at many high-altitude mainland sites.
Potentially the most powerful influence of environmental change on moorland O. brumata populations in mainland Scotland is its effect on the balance between "top-down" natural enemy pressures and altitude-related climatic severity (see Fig. 8-1). Changed patterns of November snowfall could severely affect O. brumata adult emergence and could open or close "windows" of available moorland for O. brumata colonisation. This effect may be crucial, and its future direction is unpredictable. Long-term studies of snow-lie patterns in the Scottish highlands are rare (Green, 1973; Davison, 1985; Watson et al., 1994), but their importance as indicators of climatic change has been appreciated (Watson et al., 1994). In addition, a warmer climate could permit an increase in the upper altitudinal limit of some parasitoid species, thereby reducing the lower limit of enemy-free space available to O. brumata (Figure 8-1).
In summary, the nutritional adaptability of O. brumata (Chapter 5) and the relatively unresponsive growth strategy of C. vulgaris (Aerts & van der Peijl, 1993) argue that this will not be the most intrinsically responsive insect-plant association to environmental change. Any future changes are more likely to affect the system indirectly, via altered phenological synchrony, snow-fall patterns or natural enemy abundance, than directly via altered host-plant quality.
Table 8-1. Potential effects of future environmental changes on the C. vulgaris-O. brumata interaction, and the resultant probability of outbreaks (adapted from the functional scheme devised by Landsberg & Smith, 1992). Responses shown in bold type are derived from the data in this thesis. Plain type indicates responses inferred from other sources: Kwolek & Woolhouse, 1982; Woodin et al., 1992; Iason et al., 1993; C. Gordon, pers. comm.; V.K. Brown, pers. comm. "Precipitation changes" refers to the increased winter precipitation and decreased summer precipitation predicted for the U.K. by Department of the Environment (1991). "-" indicates lack of data; "ns" indicates no significant effect. Responses in brackets indicate uncertainty or conflict of available data. (* note that there is no evidence for a direct effect of nitrogen deposition on O. brumata growth and development, acting independently of host quality).
| Increased Temperature | Increased CO2 | Increased N deposition | Precipitation Changes | Outbreak Probability | |
| Host-plant characteristics | |||||
| Growth season length | Increased | - | Increased | (Decreased) | (Increased) |
| Growth initiation date | Earlier | ns | Earlier / ns | - | Increased |
| Growth rate | ns / Increased | ns / Increased | Increased | Decreased | (Increased) |
| Nutritional quality | ns | ns / Decreased | Increased | - | (Increased) |
| Plant stress (drought) | Increased | - | (Increased) | Increased | (Increased) |
| Plant stress (frost) | Reduced | - | Increased | - | - |
| Herbivore defence (C-based) | - | (Increased) | (Reduced) | - | - |
| Herbivore characteristics | |||||
| Hatching date | Earlier | ns | ns | ns | Decreased / ns |
| Life cycle (univoltine) | ns | ns | ns | ns | ns |
| Growth and development | (Increased) | ns | ns * | - | (Increased) |
| Adult emergence success | Increased | - | - | Decreased | - |
| Natural enemy characteristics | |||||
| Predators | (Increased) | - | - | - | (Decreased) |
| Parasitoids | Increased | - | - | - | Decreased |
‡ Regrettably, no site locations or numbers of sampled larvae are given to support this data. Private correspondence between the late R.I. Lorimer and M. Shaw of Royal Museum of Scotland suggests that the 2% figure was obtained from a sample of approximately 200 larvae.
|
Previous Chapter |
 Contents Page |