|
Previous Chapter |
 Contents Page |
Next Chapter |
|
Previous Chapter |
 Contents Page |
Next Chapter |
A feeding trial was conducted to test the hypothesis that O. brumata shows compensatory feeding on Calluna vulgaris. Operophtera brumata larvae were reared on Calluna seedlings grown under raised temperature and nitrogen fertiliser treatments. Larval consumption rate was lower on nitrogen-fertilised seedlings; larval growth rate showed no response to the fertiliser treatment, whilst pupal weight was increased on nitrogen-fertilised seedlings. There was no effect of seedling growth temperature on O. brumata consumption or development. The data provide some support for the hypothesis that O. brumata shows compensatory changes in feeding rate in response to changes in diet quality; however, this mechanism was evidently not sufficient to allow O. brumata development on unfertilised seedlings to match that on fertilised seedlings.
Chapter 5 showed that O. brumata performance on C. vulgaris is remarkably unaffected by variability in host phenology and nitrogen content. This result implies a high degree of nutritional flexibility in O. brumata, and Section 5.4.2 presented the hypothesis that this flexibility is achieved by nutritional compensation. This hypothesis is tested here by a more detailed study of O. brumata feeding and development on C. vulgaris seedlings which were grown under differing conditions of temperature and nutrient availability.
Nutritional compensation is characterised by increased feeding, plus associated changes in digestive efficiency, induced in response to low food quality (Slansky & Feeny, 1977; Simpson & Simpson, 1990) (see Section 1.4). Compensation is usually detected by the use of feeding trials, which test the responses of various nutritional indices to manipulations of diet quality (e.g. Ohmart et al., 1985; Slansky & Scriber, 1989); frequently-used nutritional indices include consumption index (CI), the efficiency of conversion of ingested food to larval biomass (ECI), nitrogen utilisation efficiency (NUE), and the approximate digestibility of ingested food (AD) (Waldbauer, 1968; Slansky & Feeny, 1977).
This approach is sometimes complicated by methodological difficulties, since spurious correlations can arise between the above ratio-based indices (Raubenheimer, 1995), gravimetric errors in feeding trials may lead to artefactual response patterns (Schmidt & Reese, 1986; Simpson & Simpson, 1990), and water loss from host foliage often provides a significant source of error (Waldbauer, 1968; Scriber & Slansky, 1981). However, despite these difficulties, a commonly-recognised pattern in such studies is that ECI declines with falling dietary quality, whilst consumption rate and NUE increase (Slansky & Feeny, 1977; Ohmart et al., 1985; Simpson & Simpson, 1990). The compensatory increase in feeding rate stabilises the rate of nitrogen accumulation (Slansky & Feeny, 1977), and can result in insect growth rates that are independent of host nitrogen content (Slansky & Feeny, 1977; Ohmart et al., 1985).
In this study, the hypothesis that O. brumata development is buffered against changes in host quality by nutritional compensation was tested. Calluna vulgaris seedlings were subjected to a nitrogen fertilisation treatment, in order to increase their tissue nitrogen concentration †, thereby improving their nutritional quality for O. brumata. The effects of this assumed increase in host quality on O. brumata feeding rate and development were measured in feeding trials. A negative relationship between consumption rate and host quality would support the hypothesis that compensation had occurred. A lack of response of larval growth rate to host nitrogen-enrichment, as found in compensating insects by Slansky & Feeny (1977) and Ohmart et al. (1985), would provide further support for this hypothesis.
The Calluna seedlings were also subjected to a second treatment, raised growth temperature, applied in factorial combination with the above nitrogen treatment. Raised growth temperature was expected to produce further changes in host phenology and quality: increased temperature causes earlier budburst and accelerated development of Calluna shoots (Kwolek & Woolhouse, 1982), thereby increasing the total quantity of host foliage available for herbivores; however, under limited nutrient conditions, such increased shoot production can result in "growth dilution" of tissue nutrients, resulting in a decrease in foliage quality for herbivores (Körner, 1989; Friend & Woodward, 1990; Morecroft et al., 1992). Any responses of the O. brumata- C. vulgaris system to temperature are of particular interest, because temporal and spatial variability of temperature in the field, if it affects host quality and phenology, could also affect the potential for outbreak (see Section 1.2).
960 Calluna vulgaris seedlings, aged two years, were obtained from a recently-burned area of moorland on Balmoral Estate, N.E. Scotland (O.S. NO 29 92, altitude 400 m) on 3 and 4 November 1992. Peat was removed from an area of moorland which had not recently been burned (O.S. NO 644802, altitude 450 m) during October 1992 and passed through a 6 mm sieve whilst moist. The seedlings were established in the sieved peat in 7.5 cm diameter plastic flower pots, two seedlings per pot. Seedlings were kept in a glasshouse at 15ºC until 18 January 1993, when they were transferred to controlled-environment growth cabinets.
240 seedlings were placed in each of four growth cabinets (Fisons Fi-totron 600H). Two cabinets were set to a low temperature regime, the other two to a raised temperature regime (Table 6-1). Illumination was provided on a 24-hour light/dark cycle with a photosynthetically active radiation flux of 350 µmol m-2 s-1. Temperatures and illumination periods were adjusted during the course of the experiment in order to provide an approximate representation of seasonality (Table 6-1).
Table 6-1. Growth conditions of C. vulgaris seedlings prior to start of feeding trials.
| Date of adjustment | Light/dark cycle (hours) | Low temperature regime (light/dark temperatures ºC) | High temperature regime (light/dark temperatures ºC) |
| 18-01-93 | 8 / 16 | 8 / 6 | 11 / 9 |
| 24-02-93 | 10 / 14 | 8 / 6 | 11 / 9 |
| 24-03-93 | 12 / 12 | 8 / 6 | 11 / 9 |
| 14-05-93 | 14 / 10 | 11 / 9 | 14 / 11 |
| 26-05-93 | 16 / 8 | 11 / 9 | 14 / 11 |
Relative humidity was set to 70% in the cooler cabinets and 76% in the
warm cabinets, in order to maintain a constant daytime vapour pressure
deficit of 0.3 kPa. Plants were watered every two days with an artificial
rainwater mixture of similar pH and solute composition to rainfall in N.E.
Scotland (Edwards, 1984). Each pot received 40 ml of this mixture per week,
which constituted a background nitrogen input rate equivalent to 0.22 g
N m-2 year-1 (calculated on the basis of soil surface area in the pots).
A nitrogen fertiliser treatment was applied twice during the seedling growth period, on 14 January and 29 March 1993, to 120 randomly-selected seedlings in each cabinet. 10 ml of 43 mM aqueous ammonium nitrate solution was applied on each occasion, equivalent to an overall total application of 6.6 g N m-2 . Control pots received 10 ml of deionised water on these occasions. Control and fertilised pots were randomly arranged within each cabinet, and their positions were re-randomised after the second fertiliser addition. The lengths and fresh weights of new season's shoots were measured on 94 plants between 27 and 29 May 1993. Any near-dead or visibly-stressed seedlings were excluded from use in any of the feeding trials or measurements described here.
Adult O. brumata were collected from the moorland outbreak at Hunthill estate (Fig. 2-1ii) on 23 and 30 November 1992. Pairs of males and females were stored at a constant temperature of 5ºC in petri dishes which contained small rolls of filter paper as a substrate for oviposition. The resultant eggs were stored at 5ºC until 26 January 1993, when they were transferred to an incubator at a constant temperature of 1.5ºC in order to prevent premature hatching.
Eggs were removed from the incubator on 29 April 1993 and transferred to room temperature. Hatching occurred between 8 and 11 May 1993. Individual broods were reared in separate plastic jars, 40 cm height x 25 cm diameter, on shoots of Vaccinium myrtillus cut from turves which had been obtained from the field (O.S. NO 644802). Sixteen individual larvae from each of six broods were transferred to 96 individual plastic pots (7.5 cm height x 7.5 cm diameter with porous lids), when they had reached the third instar. Each larva was examined every day in order to determine the date of moulting to the fourth instar. Each feeding trial commenced on the date of this moult, (from 29 May to 10 June 1993) and continued for the duration of the fourth and fifth instars until pupation.
96 feeding trials were conducted simultaneously using single larvae in individual pots: two larvae from each of 6 broods were reared on seedlings from each of the eight combinations of nitrogen treatment and growth cabinet. Since larvae were initially reared on Vaccinium, they experienced a host change onto Calluna at the start of the trial. This was unavoidable, given the difficulties of rearing newly-hatched larvae on excised Calluna shoots (see Chapter 3). Pots were stored outdoors at Aberdeen in the shade (mean daily maximum temperature was 14.8ºC and mean daily minimum 8.2ºC over the trial period). Prior to use, Calluna seedlings were removed from their pots, the roots were carefully washed and covered with moist cotton wool to maintain humidity and prevent excessive water loss from the plants. Fresh seedlings were substituted every four days. Seedling fresh weight was determined before and after larval feeding, in order to determine the weight of food eaten. Due to time constraints, this weight was only measured in half of the 96 feeding pots, using three out of the overall six broods. Control seedlings were enclosed in pots without larvae in order to determine the contribution of water loss to seedling weight change in the main feeding trials. Larval fresh weight was measured daily in all pots, and frass was collected and its fresh weight determined every two days. After feeding, pre-pupal larvae were allowed to burrow into moist peat. Pupae were recovered and weighed 14 days after burrowing.
A regression of water loss on control seedling weight was used to obtain predicted water loss values for each seedling in the main feeding trials; the predicted water loss from each trial seedling was then incorporated into the following formula (from Waldbauer, 1968) to obtain corrected estimates of the weight of food eaten:
Corrected weight of food eaten = [ 1 - a/2 ] [ W - ( L + bL) ]
where
W = weight of seedling provided;
L = weight of uneaten seedling;
a = ratio of predicted seedling water loss to its initial weight
b = ratio of predicted seedling water lossto its final weight
Larval growth rate was estimated from the gradient of a linear regression of square root-transformed fresh weight on time, covering the duration of both fourth and fifth instars (see Appendix I). Nutritional indices were calculated, based on fresh rather than dry weights, as defined by Waldbauer (1968) and Slansky & Feeny (1977):
ECI (efficiency of conversion of ingested food)
= biomass gained x 100 / weight of food ingested;
CI (consumption index)
= weight of food ingested / (feeding period x mean larval weight during
trial);
CR (absolute consumption rate)
= weight of food ingested / feeding period;
AD (approximate digestibility)
= (weight of food ingested - weight of frass produced) / weight of food
ingested
Generalised linear models were fitted to data, using the GLIM (Royal Statistical Society, London, 1992) statistical package. Fitted models included terms for the effects of seedling growth temperature, nitrogen treatment, temperature-nitrogen interaction, and brood; brood effects were subsequently removed from the models, since they were never significant. Effects of nitrogen, brood and temperature-nitrogen interaction were tested for significance against an error term which described residual between-larvae variation. In order to avoid pseudoreplication (Hurlbert, 1984), the main effect of growth cabinet temperature was tested in most cases against an error term which described between-cabinet variation, and which was associated with two degrees of freedom. However, in a minority of cases where the between-cabinet error term was smaller than the between-larvae error term, testing against the latter term was considered more appropriate.
New shoot growth was first observed on seedlings in the warmer cabinets on 24 February 1993, and in the cooler cabinets on 24 March 1993. Despite this difference in the dates of growth initiation, there was no significant effect of growth temperature on mean seedling new shoot length or total new shoot length at the starting date of the feeding trials (29 May 1993) (Fig 6-1).
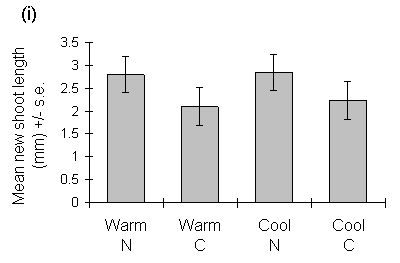
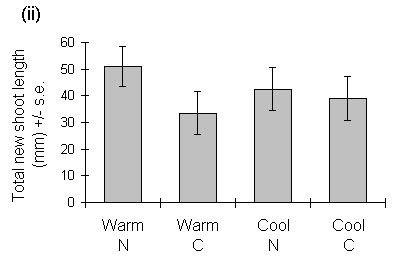
Figure 6-1. Effects of warm and cool growth regimes, and nitrogen
(N) or control (C) fertiliser treatments, on C. vulgaris shoot extension;
(i) Mean new shoot length on 29 May 1993. Effect of growth temperature:
F(1,2) = 0.085, n.s.; nitrogen treatment: F(1,88) = 5.2, p < 0.05; nitrogen-temperature
interaction: F(1,88) = 0.03, n.s. (ii) Total length of new shoots per seedling
on 29 May 1993. Effect of growth temperature: F(1,2) = 0.69, n.s.; nitrogen
treatment: F(1,88) = 3.16, n.s.; nitrogen-temperature interaction: F(1,88)
= 0.83, n.s.
However, there was a significant effect of growth temperature on the relationship between the biomass and length of new shoots (Fig. 6-2), with shoots of cool-grown seedlings less dense and visibly thinner than those on warm-grown seedlings. The total biomass of new shoot growth per seedling on 29 May, calculated from the regressions in Fig. 6-2, nevertheless showed no significant effect of growth temperature (Fig. 6-3).
Nitrogen treatment, in contrast, had no significant effect on the shoot biomass-length relationship (F(1,143) = 2.99, n.s.), but produced an increase in mean shoot extension (Fig. 6-1i). Despite the latter effect, there was no significant effect of nitrogen treatment on the total length or total biomass of new shoot growth per seedling (Figs 6-1 & 6-3).
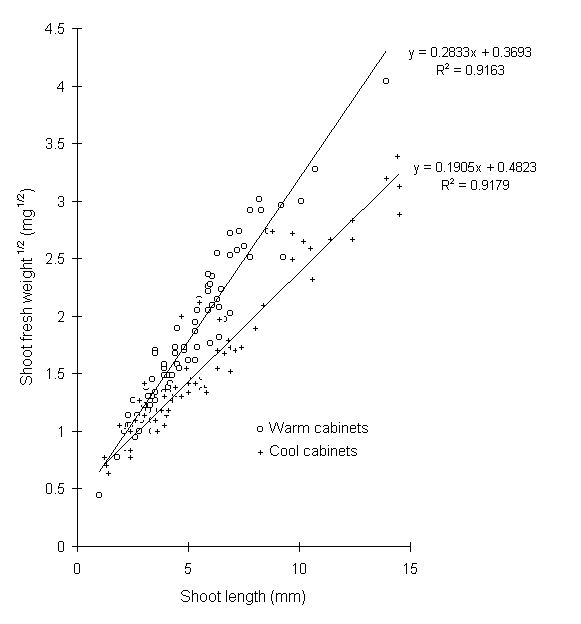
Figure 6-2. Relationship between C. vulgaris new shoot length
and square root of shoot biomass. Effect of growth temperature: F(2,144)
= 99.5, p < 0.001.
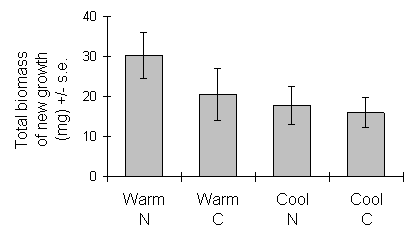
Figure 6-3. Total fresh weight of new growth per C. vulgaris
seedling on 29 May 1993. Effect of growth temperature: F(1,2) = 9.06, n.s.;
nitrogen treatment: F(1,88) = 2.89, n.s.; nitrogen-temperature interaction:
F(1,88) = 1.09, n.s.
In the control feeding pots, seedling weight change due to water loss showed a strong relationship to initial seedling fresh weight (Fig. 6-4), with lighter seedlings actually gaining in biomass over the four-day control period. This relationship was used to predict that water losses in the actual feeding trials should account for a mean of 14% of the total observed weight loss from each seedling (Fig. 6-5).
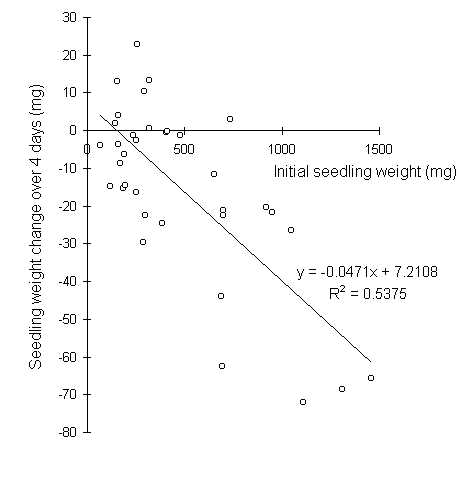
Figure 6-4. Fresh weight change of control C. vulgaris seedlings
over 4 days as a function of their initial fresh weight.
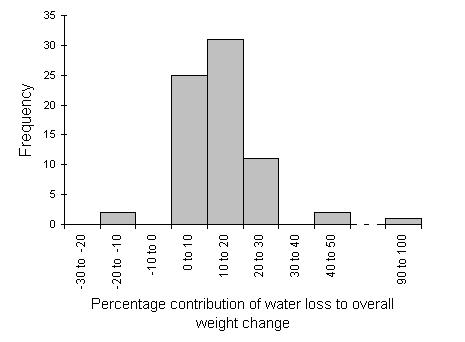
Figure 6-5. Distribution of the percentage of overall seedling
weight loss attributed to water loss rather than to larval consumption
in the feeding trial (negative values indicate an expected uptake of water
by seedlings). Mean = 14.0% s.e. = 1.62% n = 72.
Operophtera brumata development time was unaffected by seedling growth temperature in either the fourth or fifth instars (Fig. 6-6). The duration of the fourth instar was greater on nitrogen-treated seedlings, but there was no effect of nitrogen on the duration of the fifth instar (Fig. 6-6).
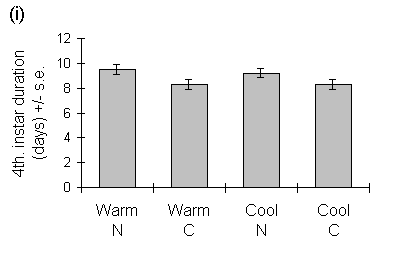
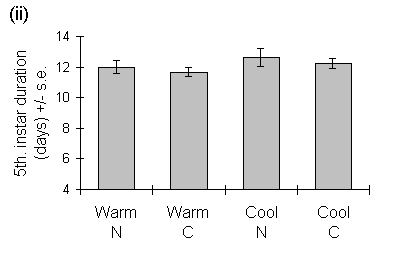
Figure 6-6. Duration of (i) fourth and (ii) fifth instars of
O. brumata reared on C. vulgaris seedlings from warm and cool growth temperatures,
with raised nitrogen (N) or control (C) treatments. Effect of seedling
growth temperature: fourth instar: F(1,2) = 0.25, n.s.; fifth instar: F(1,2)
= 2.0, n.s.; effect of nitrogen: fourth instar: F(1,69) = 8.62, p <
0.01; fifth instar: F(1,57) = 3.95, n.s.
Nitrogen treatment of seedlings significantly increased larval weight at
the moult from fourth to fifth instars, pre-pupal weight and pupal weight,
and significantly reduced the ratio of frass produced to larval biomass
gained in the fifth instar (Tables 6-2 & 6-3, Fig. 6-7). Seedling growth
temperature had no significant effect on any of these responses. There
was a significant temperature-nitrogen interaction on the ratio of frass
produced to biomass gained in the fourth instar (Table 6-3), with particularly
high production on cool-grown non-nitrogen treated seedlings. Neither ECI
nor AD indices showed any significant effect of either treatment (Tables
6-3). Large standard errors were associated with the AD calculations (Table
6-2), probably due to the combined errors of estimation of the weights
of food ingested and of frass produced. The calculated value of (weight
of food ingested - weight of frass produced) was negative in three cases,
which indicates that such errors were present. Since this term is used
as the denominator in the calculation of the ECD index (efficiency of conversion
of digested food; Waldbauer, 1968), meaningful values for that index could
not be calculated in this study.
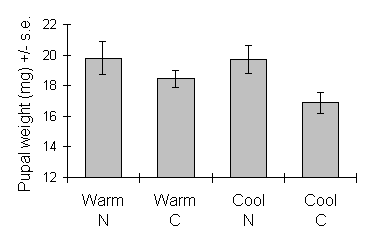
Figure 6-7. Pupal weight of O. brumata in feeding trial. Effect
of C. vulgaris growth temperature: F(1,46) = 1.71, n.s.; nitrogen: F(1,46)
= 7.90, p < 0.01; nitrogen-temperature interaction: F(1,46) = 0.93,
n.s.
Table 6-2. Performance of O. brumata larvae reared on C. vulgaris seedlings grown under warm and cool temperatures, with raised nitrogen or control treatments: table of means (and standard errors). L4 and L5 denote penultimate and final instar stages respectively.
| Temperature: |
Warm |
Cool |
||
| Fertiliser: |
+ Nitrogen |
Control |
+ Nitrogen |
Control |
| Fresh weight at moult L4 to L5 (mg) | 14.77 (0.607) | 13.65 (0.310) | 15.91 (0.533) | 12.84 (0.441) |
| Pre-pupal fresh weight (mg) | 27.02 (1.38) | 25.32 (1.45) | 26.95 (1.98) | 23.60 (1.34) |
| Frass produced / weight gained (L4) | 02.46 (0.163) | 03.00 (0.224) | 02.22 (0.210) | 03.62 (0.225) |
| Frass produced / weight gained (L5) | 18.40 (1.18) | 20.28 (0.95) | 20.21 (1.14) | 25.98 (1.38) |
| E.C.I. (L5) (%) | 04.69 (0.414) | 04.28 (0.283) | 04.34 (0.490) | 03.45 (0.258) |
| A.D. (L5) (%) | 23.76 (3.79) | 14.36 (2.48) | 12.33 (7.60) | 18.09 (3.83) |
Table 6-3. Performance of O. brumata larvae reared on C. vulgaris seedlings grown under warm and cool temperatures, with raised nitrogen or control treatments: table of significance levels.
| Effect: |
Temperature |
Nitrogen |
Temp . Nitrogen |
| Fresh weight at moult L4 to L5 | n.s. | F(1,60) = 19.17 *** | n.s. |
| Pre-pupal fresh weight | n.s. | F(1,59) = 7.56 ** | n.s. |
| Frass produced / weight gained (L4) | n.s. | F(1,60) = 23.81 *** | F(1,60) = 4.43 * |
| Frass produced / weight gained (L5) | n.s. | F(1,54) = 8.36 ** | n.s. |
| E.C.I. (L5) | n.s. | n.s. | n.s. |
| A.D. (L5) | n.s. | n.s. | n.s. |
Consumption index (CI) in the fifth instar was significantly lower on nitrogen-treated seedlings than on controls, but was not affected by seedling growth temperature (Fig. 6-8).
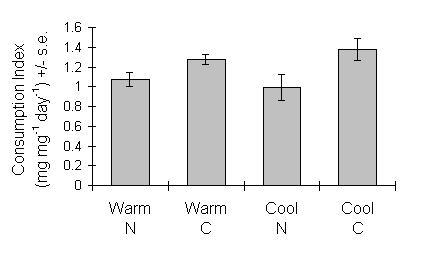
Figure 6-8. Consumption index for final instar O. brumata. Effect
of C. vulgaris growth temperature: F(1,2) < 0.01, n.s.; nitrogen: F(1,26)
= 12.97, p < 0.01; nitrogen-temperature interaction: F(1,26) = 0.21,
n.s.
Consumption index showed no relationship to larval fresh weight at the start of the feeding trial (Fig. 6-9i), but was significantly negatively correlated with larval weight at the moult from fourth to fifth instars (Fig. 6-9ii), with pre-pupal weight (Fig. 6-9iii) and with pupal weight (Fig. 6-9iv).
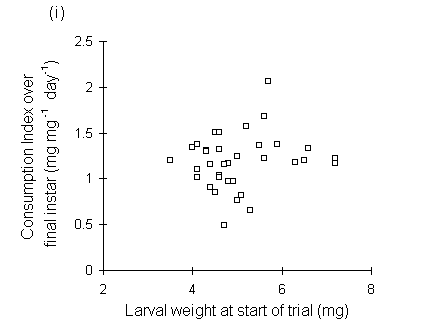

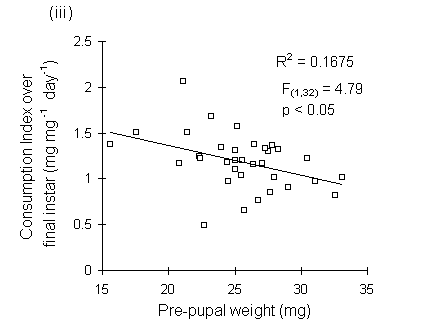
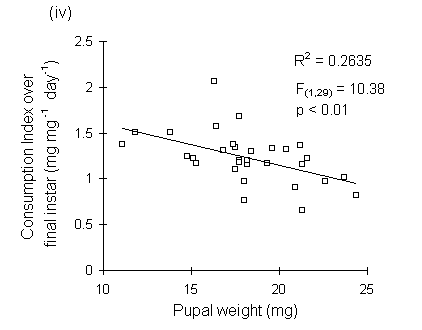
Figure 6-9. Relationship between O. brumata consumption index (CI) in final instar and: (i) larval weight at start of feeding trial; (ii) larval weight at moult from fourth to fifth instar; (iii) pre-pupal weight; (iv) pupal weight. Significance levels for regressions are shown in (ii) to (iv); the relationship in (i) is non-significant.
Absolute consumption rate was also lower on nitrogen-treated seedlings (Fig. 6-10), but showed no significant relationship to larval, pre-pupal or pupal weights (Fig. 6-11).
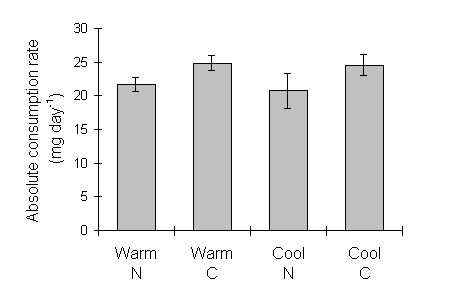
Figure 6-10. Absolute consumption rate (CR) for final instar
O. brumata. Effect of C. vulgaris growth temperature: F(1,2) = 0.3, n.s.;
nitrogen: F(1,29) = 4.9, p < 0.05; nitrogen-temperature interaction:
F(1,26) = 0.03, n.s.
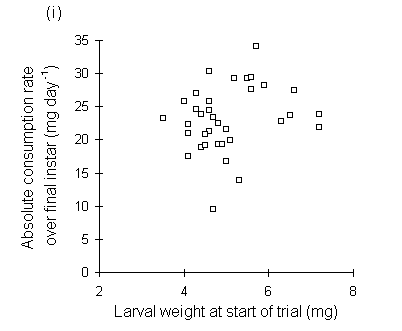

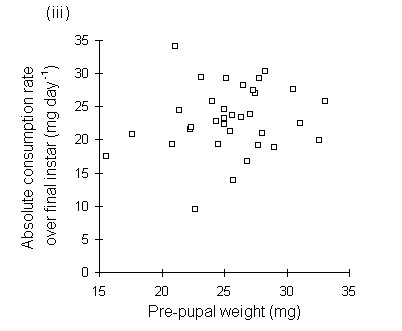
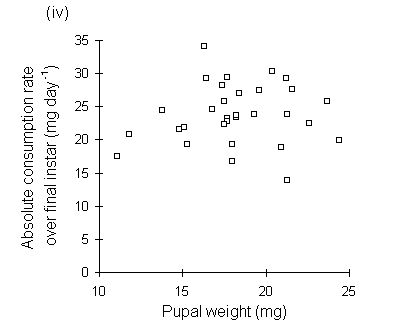
Figure 6-11. Relationship between O. brumata absolute consumption rate (mg food eaten day-1) in final instar and: (i) larval weight at start of feeding trial; (ii) larval weight at moult from fourth to fifth instar; (iii) pre-pupal weight; (iv) pupal weight. All relationships are non-significant
Larval growth rate over the fourth and final instars was not significantly affected by either experimental seedling treatment (Fig. 6-12).
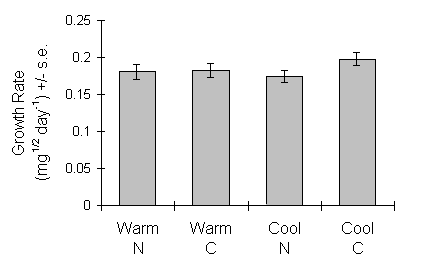
Figure 6-12. Growth rate of O. brumata on C. vulgaris seedlings (fourth and fifth instars combined). Effect of seedling growth temperature: F(1,2) = 0.16, n.s.; nitrogen treatment: F(1,66) = 2.53, n.s.; nitrogen-temperature interaction: F(1,66) = 1.47, n.s.
This study supports the hypothesis that O. brumata shows compensatory changes in feeding rate in response to experimental changes in C. vulgaris nutritional quality. Larval consumption index (CI) in the fifth instar was significantly lower on nitrogen-treated seedlings than on controls (Fig. 6-8), and absolute consumption rate was similarly lower on nitrogen-treated seedlings (Fig. 6-10).
Furthermore, larval growth rate was unaffected by the experimental seedling growth treatments (Fig. 6-12). This is again consistent with the expected effects of compensatory feeding (Slansky & Feeny, 1977; Ohmart et al., 1985), and so supports the above conclusion.
However, O. brumata development was not completely insensitive to the experimental treatments; there is strong evidence in Tables 6-2 and 6-3 that nitrogen-enrichment of Calluna seedlings improved their nutritional quality (since larval frass production per unit biomass gain was reduced on nitrogen-enriched seedlings), and resulted in increased larval, pre-pupal and pupal weights. Compensatory changes in feeding rate were, therefore, evidently not sufficient to raise O. brumata developmental performance on unfertilised seedlings to the same level as that on fertilised seedlings. The data, therefore, support the view that compensation did occur, but that it did not totally eliminate the effects of host quality on performance.
The significant negative relationships shown between CI and larval, pre-pupal and pupal weights (Fig. 6-9) initially appear to provide further evidence for a negative relationship between consumption and host quality ‡. However, no negative relationships were found between absolute consumption rate and the same weight parameters (Fig. 6-11). The significant negative relationships shown in Fig. 6-9 are, therefore, probably artefacts of the fact that CI is a "weight-relative" index, standardised to mean larval body weight (Waldbauer, 1968). Any measurement which correlated positively with mean larval body weight during the trial would automatically be expected to correlate negatively with CI.
The potential for such spurious auto-correlation when using ratio-based nutritional indices is large (Raubenheimer, 1995), and has been widely recognised (Packard & Boardman, 1988; Raubenheimer, 1995). Slansky & Feeny (1977) relied primarily on absolute consumption rate in order to demonstrate compensation, and Ohmart et al., (1985) argued that the use of the weight-relative CI "masked relationships that were clearly demonstrated by absolute measures"; Slansky & Wheeler (1991) were more fortunate in being able to demonstrate equivalent compensatory responses in both absolute and weight-relative consumption indices. In the present study, the fact that both absolute and weight-relative consumption rates showed similar responses to the seedling nitrogen treatment (Figs 6-8 & 6-10) proves that this effect is not simply an artefact of greater larval weights on nitrogen-treated seedlings; the conclusion that O. brumata showed compensatory feeding is, therefore, safe from this criticism.
Seedling growth temperature had no significant main effect on any aspect of O. brumata performance (Table 6-3; Figs 6-6 to 6-12). This is surprising, since raised growth temperature had a strong effect on shoot morphology (Fig. 6.2), and advanced budburst date by one month. However, any effect of temperature on seedling quality was evidently small in comparison to the nitrogen treatment, and of minimal consequence for O. brumata development. This result argues that temperature variation in natural systems, for example due to altitude or future climatic change (Vitousek, 1992), is unlikely to affect the nutritional relationship between O. brumata and C. vulgaris. This contrasts with the potentially strong direct effects of temperature on O. brumata life history discussed previously (Chapters 3 & 4). However, feeding studies in the field would be necessary to confirm these results under more realistic temperature and light regimes.
The positive effects of seedling nitrogen treatment on larval, pre-pupal and pupal weights (Table 6-2; Fig. 6-7) were the result of increased feeding times (Fig. 6-6), not increased growth rates (Fig. 6-12). The benefits of such a response in the field are questionable, since prolonged feeding will carry increased risks of parasitism and predation (Cheng, 1970; Picozzi, 1981; Loader & Damman, 1991). Larval development in plastic pots must be expected to deviate somewhat from the equivalent responses when feeding ad libitum.
The positive effect of Calluna nitrogen treatment on O. brumata pupal weight implies that nitrogen enrichment of moorlands (Pitcairn et al., 1995) can improve O. brumata reproductive potential. This is consistent with the hypothesis that outbreaks are caused by enrichment (Chapter 1), but contradicts the conclusion of Chapter 5, that O. brumata development is insensitive to Calluna nitrogen concentration. However, the nitrogen treatment applied here was large in comparison to the inputs commonly experienced by Calluna in the field (Pitcairn et al., 1995). Although time prevented the acquisition of Calluna tissue chemistry data in this case, it is possible that the nitrogen treatment used in this study caused a greater change in nitrogen concentration than the natural decline of 15% investigated in Chapter 5 (Fig. 5-7). The extent to which compensation mechanisms can buffer development against host quality will obviously depend on the magnitude and direction of the quality changes considered.
This chapter provides partial support for the argument advanced in Section 5.4.2, that nutritional flexibility and compensatory feeding are fundamental characteristics of O. brumata nutrition on Calluna. There is clear evidence that compensatory feeding did occur, but its effect was not sufficiently strong to allow performance on unfertilised seedlings to match that on fertilised seedlings. In the terminology of the model in Figure 5-9, the gradient of the response of performance to quality was greater than zero in this case. The extent to which O. brumata is sensitive to variability in host quality in the field is difficult to assess from these results; more information about the range of Calluna tissue nitrogen concentrations experienced in the field, as well as in manipulative feeding trials, is clearly required (see Chapter 7). However, the lack of response to seedling growth temperature shown here, despite changes in plant phenology, argues that O. brumata did exhibit appreciable nutritional flexibility.
† See Chapter 7 for confirmation that nitrogen fertilisation of Calluna does produce an increase in tissue nitrogen concentration (see also Iason et al., 1993; Hartley & Gardner, 1995).
‡ since pupal weights in particular are commonly used as good indicators of host quality (e.g. Feeny, 1970).
|
Previous Chapter |
 Contents Page |
Next Chapter |