|
Previous Chapter |
 Contents Page |
Next Chapter |
|
Previous Chapter |
 Contents Page |
Next Chapter |
Differences in hatching date were demonstrated between moorland and oak woodland O. brumata populations, and moorland populations showed close synchrony between hatching and C. vulgaris budburst date. However, experimental delay of hatching date had no adverse effects on O. brumata performance on C. vulgaris. There is, therefore, no evidence that moorland O. brumata populations benefit from hatching in synchrony with C. vulgaris budburst. Differences in hatching date between populations are more likely to be caused by adaptations of egg development to local temperature regimes. A moorland O. brumata biotype showed better survival on Calluna than an oak-feeding biotype; however, it is unclear whether this difference was due to specialisation of the moorland biotype to Calluna, since other performance indicators showed no significant difference between biotypes.
Chapter 4 demonstrated life history adaptation of a moorland O. brumata population to its climatic environment, but found no evidence that moorland populations are nutritionally specialised to Calluna vulgaris. These conclusions are re-examined in the present chapter, which focuses on the importance of O. brumata egg hatch date for subsequent performance on Calluna. Two hypotheses are tested: (i) that temporal synchrony between O. brumata hatching and Calluna budburst is an important determinant of larval survival and development on this host; and (ii) that moorland outbreak O. brumata populations show adaptations which enable them to attain this synchrony. In order to test these hypotheses, the life histories and performance on Calluna of moorland and oak woodland O. brumata populations were contrasted. The effects of an artificial hatch delay were investigated, and a model is presented to explain the apparent lack of sensitivity of O. brumata to Calluna phenology.
Many outbreaking species of phytophagous insects emerge and begin feeding in synchrony with the budburst of their hosts (Nothnagle & Schultz, 1987; Hunter, 1991) (see Section 1.2.4). This enables larvae to feed on newly-flushed leaves, which are generally nutritionally superior to older foliage (Feeny, 1970; Schweitzer, 1979; Larsson & Ohmart, 1988). Hatch-budburst synchrony is an important determinant of larval performance and overall defoliation levels for a range of forest Lepidoptera (Raupp et al., 1988; Du Merle, 1988; Quiring, 1994; but see Crawley & Akhteruzzaman, 1988).
Several studies in forest systems have tested the effects of host phenology on O. brumata, but have produced conflicting results. On broadleaved trees such as Quercus spp. (oak) and Malus spp. (apple), synchrony between hatching and budburst can affect O. brumata population densities (Varley & Gradwell, 1968; Holliday, 1977). Feeny (1970) showed that oak foliage declines in nutritional quality for O. brumata as it matures, whilst Wint (1983a) showed that attainment of close hatch-budburst synchrony improved O. brumata pupal weight on Quercus robur. However, Watt & McFarlane (1991) found that the requirement for hatch-budburst synchrony was less strong for O. brumata on Picea sitchensis (Sitka spruce), a recently colonised host, than on broadleaved species. The importance of host nutrient status is also less clear for O. brumata on spruce (Hunter et al., 1991). These conflicting results were reviewed by Hunter (1992), who argued that the relationship between host phenology and insect herbivory varies between sites and between hosts, possibly due to differences in plant spacing, and in the evolutionary ages of the insect-plant associations in question.
If hatch-budburst synchrony can be shown to be an important determinant of O. brumata performance on C. vulgaris, then this will strengthen the hypothesis that moorland O. brumata populations are sensitive to, and potentially constrained by, the nutritional quality of this host (see Chapters 1 & 3 for development of this argument). In order to determine whether this is the case, an experiment was conducted to test the effect of artificially delaying O. brumata hatch date on the quality of available Calluna foliage, and on the survival and development of O. brumata on this foliage.
The need to synchronise development to the phenology of a specific host can result in local adaptation of an insect population, such that it differs in hatching date and genetic constitution from conspecific populations on other hosts (Mitter et al., 1979; Komatsu & Akimoto 1995) (Section 1.2.4). In order to determine whether this is the case for O. brumata on Calluna, hatching date was determined in situ for a moorland outbreak population; the hatching dates of moorland and oak woodland O. brumata populations were contrasted, and the implications of the observed differences for survival and development on Calluna were investigated.
Adult O. brumata were captured from the Hunthill moorland outbreak (see Section 2.3.2) during November 1993. Male and female moths were placed in pairs into 7 cm diameter plastic pots, which were tied securely at ground level onto wooden fence posts at the site, and were sheltered from direct sunlight by stones. Small rolls of filter paper were provided in the pots as an oviposition substrate. Between late November and late March 1994, the pots were buried beneath deep snow which covered the entire site (see Fig. 3-10ii). On 12 April 1994, eggs were removed from the pots, and those which were firmly attached to the filter paper were counted. In total, 68 eggs were obtained from six different broods. The eggs were placed inside fine-mesh "Organza" polyester bags which were securely tied onto the wooden posts at 20 cm above ground level. Previous studies had shown that the warming effect of these bags was negligible (J.E. Kerslake, unpublished data). Three such bags were used, and the eggs were distributed approximately evenly between them, with no attempt to keep broods separate. A further four O. brumata eggs were found in crevices in the wooden fence posts at the site, and these were marked and added to the study. Darkening (which indicates imminent larval emergence) and hatching of eggs was monitored at the site on seven occasions between 19 April and 16 June 1994. The length of new season's shoots of C. vulgaris was also recorded at six randomly-selected points within the site on each of these monitoring dates.
Stocks of O. brumata larvae were obtained in 1994 from three different outbreak populations: Hunthill high-altitude moorland (Fig. 2-1ii), Swartaback low-altitude moorland on Orkney (Fig. 2-1i), and Bonar Bridge oak woodland (Fig. 2-2i). Larvae were reared to pupation on the hosts from which they were obtained, and pupae were stored under uniform conditions (see Section 4.2 for full method). Emergent adults were mated only with others of the same population, in plastic jars which were stored in a growth cabinet at a constant temperature of 5ºC. Eggs were laid onto small rolls of filter paper which were provided in the jars. Eggs were stored at a constant temperature of 5ºC from the time they were laid, and their patterns of hatching were monitored every 6 days between 17 March and 12 May 1995. A total of 655 eggs from 55 broods were monitored: 30 broods from Hunthill, 7 from Swartaback and 18 from Bonar Bridge.
Thirty-six Calluna vulgaris plants (approx. 80 cm in diameter) were obtained from an upland moor (O.S. NO 644802, altitude 450 m) and established in an outdoor plot at Aberdeen in October 1994. The roots of the plants were isolated from the surrounding soil of the plot by plastic sheeting. Eighteen of these plants were assigned at random to a "delayed hatch" treatment (described below), and eighteen to "undelayed hatch".
Eighteen broods of O. brumata eggs were obtained from moorland outbreak populations, and eighteen from oak (Quercus spp.) woodland populations. These will be referred to here as "moorland" and "oak" biotypes respectively. Ten of the moorland broods were laid by adult moths captured at the Hunthill outbreak during November 1994; the remaining eight moorland broods derived from larvae taken from the Swartaback outbreak on Orkney in June 1994, which were subsequently reared to adults and then mated together. Sixteen of the oak-feeding broods were obtained from larvae taken from the Bonar Bridge oak outbreak in June 1994, reared to adults and mated together; the remaining two oak broods were obtained from adult females captured at Wytham Woods near Oxford in November 1994 (L. Cole, pers. comm.).
Each moorland brood was assigned at random to one "undelayed hatch" heather plant. This procedure was then repeated with the oak broods. Each "undelayed hatch" plant was therefore allocated for use by both one moorland and one oak brood. The same pairs of broods were then also allocated to corresponding "delayed hatch" plants. This experimental design was chosen in order to permit direct paired comparisons between the performance of oak and moorland O. brumata broods on the same individual Calluna plant. Variation in larval performance due to differences between individual plants could therefore be eliminated from comparisons between O. brumata populations. This experimental design is illustrated schematically in Figure 5-1.
Figure 5-1.
Design of feeding trial to compare performance of "oak" and "moorland"
O. brumata biotypes on C. vulgaris, with delayed and undelayed hatch treatments.
Ellipses represent individual C. vulgaris plants; rectangles represent
feeding bags containing eggs of either oak (O) or moorland (M) O. brumata
biotype.
Eggs from each brood were placed onto their allotted Calluna plants as follows:
For the "undelayed hatch" treatment, eggs were stored at a constant temperature of 5ºC from the time they were laid until 9 February 1995, when they were placed onto their allotted plants in 50 cm x 30 cm Organza polyester feeding bags. Each feeding bag was placed over approximately 15 green Calluna shoots, and was sealed with wire tied tightly around the base of the woody stems. Each feeding bag contained 12 eggs from a single brood, and each plant therefore received two feeding bags. Care was taken to ensure that these bags were widely separated from one another on the plant, and that they contained approximately equal amounts of foliage.
In order to monitor egg viability and hatch dates for each brood, a second pair of smaller Organza bags were tied onto each plant, containing eggs from the same two broods used in the main feeding bags. These smaller "control" bags enclosed no foliage, and each contained ten O. brumata eggs attached to small pieces of card by a light water-soluble gum. Previous pilot experiments had indicated that the use of this gum did not adversely affect hatching. Small tin-foil rain shields were tied above the control bags in order to prevent the eggs from drowning or becoming detached from the card. Darkening and hatching of the control eggs was monitored weekly from 21 March onwards, and the length of new season's growth on a tagged shoot of each plant was also measured on these occasions.
For the "delayed hatch" treatment, the remaining eggs from the same broods were stored at 5ºC from the time they were laid until 17 March 1995. On this date they were transferred to a refrigerator at 1ºC in order to delay their hatching. They remained at 1ºC until 1 May 1995, when they were brought out onto the experiment. Pairs of broods were placed in feeding bags onto their allotted "delayed hatch" Calluna plants in the manner described above for the "undelayed" treatment. Twelve eggs were used in each feeding bag, as before. Control bags were set up as previously described, and all plants were regularly monitored as before.
Since broods of eggs hatched on different dates, it was necessary to standardise the length of feeding time experienced by each set of larvae prior to harvest. This was done by harvesting each feeding bag 42 days after the estimated date of hatching of the eggs inside it. The estimated hatching date was calculated for each feeding bag as the interpolated hatch date of the median viable egg from each corresponding control bag.
At harvest, surviving larvae were removed from the feeding bags, weighed, and their instar stage determined by head capsule width (Cuming, 1961). Larvae were then allowed to complete their development to pupation in 7 cm diameter plastic pots which also contained a 3 cm-deep layer of damp moorland peat mixed with an inert water-retaining polyacrylamide gel. Larvae from each feeding bag were reared in separate pots. Fresh cut shoots from the appropriate C. vulgaris plant were supplied to each pot every 48 hours. Pots had porous lids, and were stored in a shaded location at approximately 12ºC. Pupae were sieved out of the peat nine weeks after harvest date, weighed and sexed.
Samples of foliage were taken from each Calluna plant in the above feeding trial (Section 5.2.3) on the harvest dates of each of its associated feeding bags. Each plant was therefore sampled on two occasions. Shoots were selected close to but not including those shoots which were contained within the corresponding feeding bag, in order to prevent contamination from frass. Shoots were excised at the base, the new season's growth was dissected from them, washed with deionised water and dried for a week at 70ºC. Samples were ground in a ball mill, and re-dried prior to analysis. Total nitrogen and carbon concentrations were determined using a Fisons NA 1500 NCS autoanalyser, calibrated with a bulked soil standard. Vinyl gloves were worn during all stages of this protocol.
All data was analysed using the GLIM (Royal Statistical Society, London, 1992) statistical package. Survival analysis was used to test the effect of source population on O. brumata hatching date (Crawley, 1993). In the experimental feeding trial (Section 5.2.3), between-plant variability was separated from within-plant variability in order to obtain appropriate error terms for testing the main effects of hatch delay treatment and biotype respectively. For analysis of larval survival, the conditional probability of larval survival given successful hatching was calculated for each feeding bag as follows:
P (survival given hatching) = P (hatch and survive) / P (hatch)
P (hatch and survive), the probability of survival from egg to larval harvest, was obtained from the feeding bags; P (hatch), the probability of egg hatching, was obtained from the control bags. A binomial model was then fitted to this data set.
Larval growth rate was calculated as untransformed weight gain per day (see Appendix I).
Mean larval developmental stage was described by a development index, as used previously (Chapter 3), calculated as follows:
Development index = ![]() where i = instar stage and pi = proportion of
larvae in instar i.
where i = instar stage and pi = proportion of
larvae in instar i.
This response was analysed using a binomial model.
Although spring temperatures in 1994 were below average in N.E. Scotland (Cullum, 1994), there was a close synchrony between O. brumata hatching (Fig. 5-2i) and the initiation of new growth by C. vulgaris (Fig. 5-2ii).
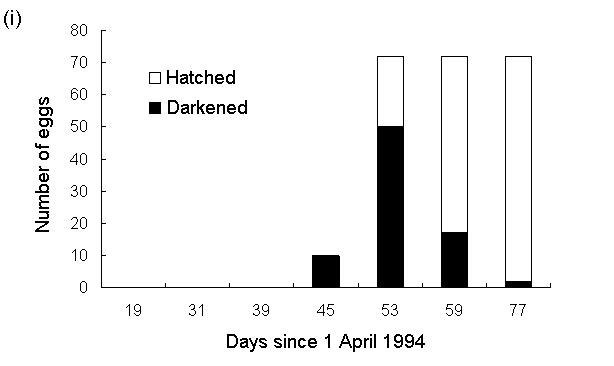
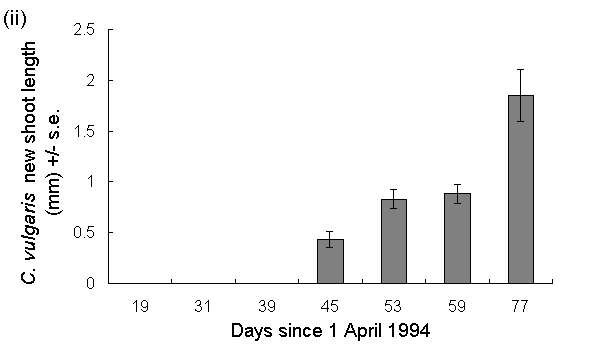
Fig. 5-2. (i): Hatching pattern of O. brumata and (ii):
shoot length of C. vulgaris in situ at Hunthill moorland outbreak site,
1994.
By 29 May, the majority of the eggs had hatched, and the new season's growth of C. vulgaris was approximately 1 mm long (Fig. 5-2, day 59). This was also the first date in 1994 on which first and second instar O. brumata larvae were observed on the surrounding vegetation (J.E. Kerslake, pers. obs.), which suggests that the hatching dates obtained in this study were similar to those of eggs laid naturally at the site.
At a constant temperature of 5ºC, egg hatch commenced on 17 March 1995 and was monitored until 12 May 1995. During this time, 96.4% of eggs from the Hunthill population, 85.7% of eggs from Swartaback, and 59.2% of eggs from Spinningdale hatched (Fig. 5-3). Further eggs may have hatched after this date, particularly in the Swartaback population, and there is consequently a degree of censoring in the data, which was taken into account in the subsequent survival analysis (Crawley, 1993).
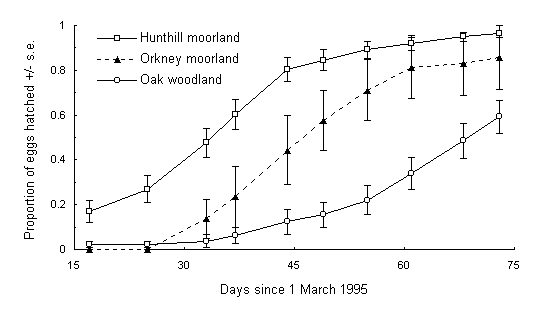
Fig. 5-3. Hatching patterns of O. brumata from Hunthill moorland,
Orkney moorland and Bonar Bridge oak woodland populations at constant 5ºC.
Hatching is expressed as the cumulative proportion of the total number
of eggs monitored during the study period of 17 March to 12 May 1995, taking
broods as replicates.
Source population had a significant effect on hatching date (F(1,52) =
5.13, p < 0.05) (Fig. 5-3). Mean hatching date was 4 April for eggs
from the Hunthill high-altitude moorland population, 13 April for the Orkney
moorland population, and 28 April for the Bonar Bridge oak-feeding population.
Hatching dates in the feeding trial showed a similar pattern to that seen under constant temperature conditions: the undelayed moorland O. brumata broods hatched approximately 16 days in advance of the undelayed oak-feeding broods (Fig. 5-4). The hatch delay treatment succeeded in postponing emergence of both biotypes by approximately 30 days, and so preserved the difference in hatching dates between biotypes (Fig. 5-4).
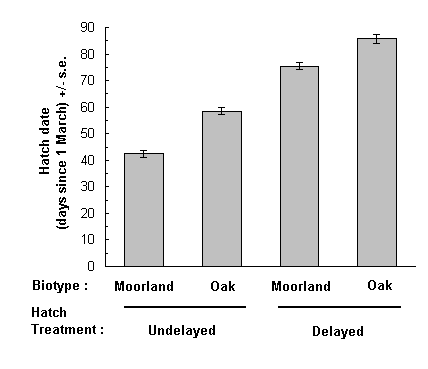
Fig. 5-4. Hatching dates of eggs from moorland and oak-feeding
biotypes of O. brumata in the feeding trial. Delayed and undelayed hatching
treatments are indicated. Hatching date was calculated as the interpolated
date of hatching of the median viable egg from each brood.
There was, therefore, a steady progression in hatching dates over the four
treatment-biotype combinations. This was reflected in a steady increase
in the lengths of Calluna new season's shoots available to newly-hatched
larvae over these four combinations, and in the lengths of these shoots
42 days after hatching, when larvae and shoots were harvested (Fig. 5-5).
Larvae in each treatment-biotype combination therefore fed on Calluna
over a different range of its seasonal development.
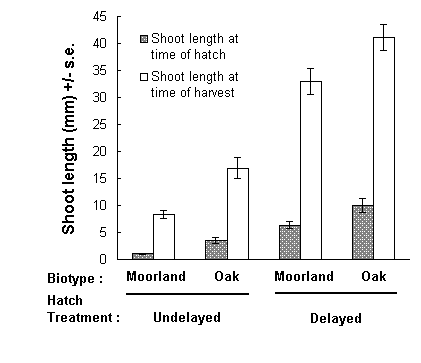
Fig. 5-5. Length of current season's shoots of C. vulgaris at
time of hatching and time of harvesting of each treatment-biotype combination.
Delay of hatching had no effect on the probability of larval survival in either moorland or oak biotypes (Fig. 5-6). However, there was a highly significant biotype effect on survival: oak biotypes achieved considerably lower survival rates on C. vulgaris than moorland biotypes (Fig. 5-6).
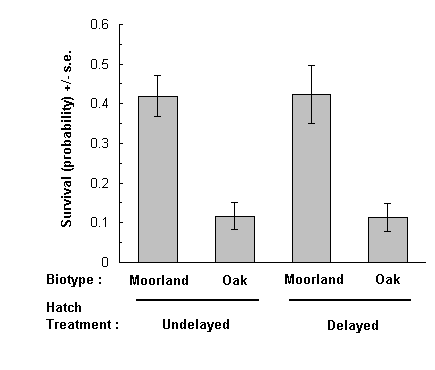
Fig. 5-6. Conditional probability of larval survival given successful
hatching on C. vulgaris for each treatment-biotype combination (standard
errors are taken directly from the data, not from the logit link model)
(Effect of biotype: F(1,32) = 57.6, p < 0.001 ; effect of hatch
delay: F(1,34) < 0.01, n.s.).
The above effect of biotype on survival was observed both in undelayed and delayed hatching treatments (Fig. 5-6), and was therefore not related to the difference in hatching dates between biotypes. Furthermore, the effect was not attributable to differences in egg viability between biotypes, since the analysis of survival as a conditional probability corrects for this factor (see above), and there was in any case no significant effect of biotype on egg viability (F(1,34) = 0.62 , n.s.).
A potential difficulty in the interpretation of the biotype effect stems from the fact that moorland and oak biotype feeding bags were not strictly independent of one another, since they were placed in pairs on individual plants. It could be argued that the low survival of the oak biotype was due to induced defensive responses in the plants to damage from the earlier-hatching moorland biotype in the other bags. Induced biochemical changes in response to herbivore damage have been documented in a range of plant species (Raupp & Denno, 1984; Haukioja & Hanhimäki, 1985; but see Fowler & Lawton, 1985). Whilst it is not possible to fully reject this explanation, bags were as widely spaced as possible on the plants (at least 50 cm), the plants were large, and the overall level of herbivory light (less than 5% of the total foliage of each plant was enclosed in the feeding bags). In a small number of cases where no larvae were found in the moorland biotype bags due to poor egg viability, the survival of associated oak broods was still poor. Interaction between feeding bags is therefore unlikely to have been a major cause of the observed difference in survival rates. Differing responses between biotypes to the cold storage of eggs prior to the start of the experiment may also have affected survival rates, although since there was no evidence for differences in egg viability between biotypes, any such response could only have affected post-hatching larval viability.
There was no significant effect of biotype on larval growth rate or development index after 42 days, but the hatch delay hatch treatment resulted in a highly significant increase in both of these responses (Tables 5-1 & 5-2). Potential causes of this unexpected result include warmer weather conditions during the later larval feeding period (Myers, 1992), and the availability of a larger quantity of new season's growth of Calluna shoots in the feeding bags (although care was taken to enclose large quantities of foliage in the feeding bags for both hatching treatments, and there was no indication that foliage was limiting at any time during the course of the experiment). Pupal weight showed no significant effect of either biotype or hatch delay (Tables 5-1 & 5-2).
Table 5-1. Performance of O. brumata moorland and oak biotypes on C. vulgaris, with undelayed and delayed hatching treatments: table of means (and standard errors).
| Treatment: |
Undelayed |
Delayed |
||
| Biotype: |
Moorland |
Oak |
Moorland |
Oak |
| Growth rate (x 10) (mg day-1) | 4.03 (0.310) | 3.96 (0.863) | 7.11 (0.457) | 8.38 (0.910) |
| Development index | 4.33 (0.124) | 4.14 (0.298) | 4.82 (0.892) | 4.81 (0.188) |
| Pupal weight (mg) | 23.70 (0.52)0 | 21.05 (1.17)0 | 24.08 (0.685) | 24.51 (1.26)0 |
Table 5-2. Performance of O. brumata moorland and
oak biotypes on C. vulgaris:
table of significance levels (*** indicates p < 0.001).
| Effect: |
Hatch delay |
Biotype |
Delay . Biotype |
| Growth rate (x 10) | F(1,32) = 19.53 *** | n.s. | n.s. |
| Development index | F(1,32) = 15.55 *** | n.s. | n.s. |
| Pupal weight | n.s. | n.s. | n.s. |
Analysis of new season's growth showed a highly significant drop in C. vulgaris tissue nitrogen concentration over the course of the harvesting period (Fig. 5-7i). Total nitrogen content fell by approximately 15% between 15 May and 2 July 1995, whilst total carbon content showed no significant trend over that time (Fig. 5-7ii). Date of harvest accounted for 23% of the variation in nitrogen content (Fig. 5-7i), although residual variability between individual plants accounted for a further 57.3%. Both the delayed hatch treatment and the later hatching of oak biotype eggs resulted in the corresponding larvae experiencing foliage with a significantly lower nitrogen content than the undelayed hatch or moorland biotype treatments respectively (hatch delay: F(1,34) = 7.73, p < 0.01; biotype: F(1,34) = 4.1, p < 0.05). The absolute values of nitrogen concentration obtained here were high in comparison with those from other studies (Pitcairn et al., 1995; Hartley & Gardner, 1995), probably due to the high sensitivity of the assay technique used here (see Appendix II). Regression analysis showed no significant relationship between C. vulgaris nitrogen content and either larval survival (F(1,34) = 1.96, n.s.), larval growth rate (F(1,32) < 0.01, n.s.) or pupal weight (Fig. 5-8).
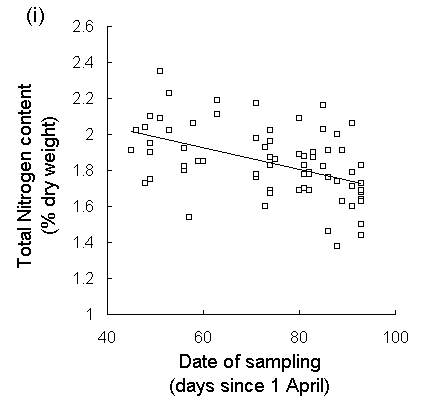
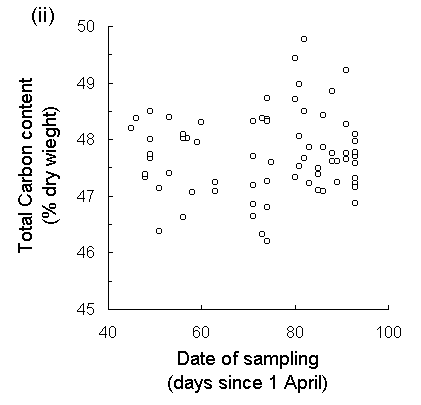
Fig. 5-7. (i) Total nitrogen concentration and (ii) total carbon concentration in new season's shoots of C. vulgaris, plotted against date of harvest. (Nitrogen: F(1,70) = 20.97, p < 0.001, R2 = 0.23; Carbon: F(1,70) = 0.04, n.s.)
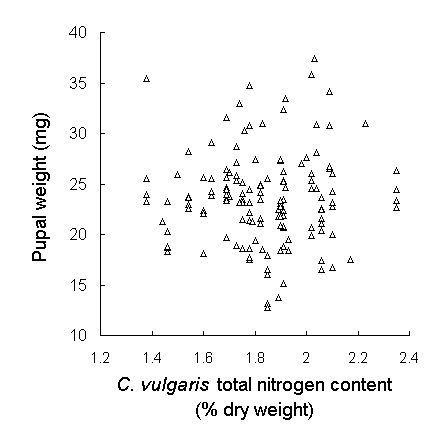
Fig. 5-8. Relationship between O. brumata pupal weight
and total nitrogen content of C. vulgaris new season's shoots
at the time of harvest (F(1,32) = 0.023, n.s.)
This study demonstrates that temporal synchrony of O. brumata hatching with Calluna budburst is of little importance for larval survival and development on this host. Delaying hatch by a month had no adverse effect on any of the indicators of larval performance measured, despite falling nitrogen concentration in the available Calluna foliage over this time. Operophtera brumata demonstrated a remarkable ability to maintain adequate larval development on a food resource of steadily declining quality.
These results are similar to previous work by Myers (1992), who showed that a delay of three to six weeks in hatching caused no reduction in survival or development of Malacosoma californicum pluviale Dyar (western tent caterpillar) on Alnus rubra Bong (red alder) and Malus diversifolia (crab apple). However, the results of the present study contrast strongly with much previous work which has stressed the importance of hatch-budburst synchrony for O. brumata performance on broadleaved hosts (see Section 5.1.2). Even on a conifer, Sitka spruce, where hatch-budburst synchrony is less important for O. brumata, larval survival still declines rapidly after budburst as the foliage ages (Watt & McFarlane, 1991). This chapter therefore highlights an apparent paradox, namely that O. brumata development displays differing sensitivities to plant phenology and nutritional quality on different host species.
One hypothesis to explain the unimportance of hatch-budburst synchrony shown here is that Calluna does not decline in quality sufficiently rapidly to make this effect detectable. Although this study demonstrated a highly significant drop of 15% in mean total nitrogen content in C. vulgaris over 50 days (Fig. 5-7), Wint (1979) found that nitrogen concentrations in oak foliage declined by approximately 40% over the same time period. The slower decline in C. vulgaris nitrogen content may reduce the impact of plant phenology on larval development. In addition, there is considerable variability in nitrogen content between individual C. vulgaris plants (Fig. 5-7i), which will have limited the ability of this study to resolve main effects of hatch delay. Furthermore, an increase in ambient temperatures over the study period may have improved the performance of larvae in the delayed hatch treatment, outweighing any negative effects of reduced host quality (Myers, 1992).
However, it is important to recognise that Calluna tissue nitrogen concentrations are substantially lower than those of many broadleaved plants (Wint, 1979). Calluna is a demonstrably sub-optimal host for O. brumata (Chapter 3), and a drop of 15% in total nitrogen content should have particularly severe implications for larval development on a host that is already nutrient-poor. The lack of any such negative effect, therefore, prompts an alternative explanation, that O. brumata is genuinely insensitive to Calluna nitrogen content, and that moorland outbreaks are not a response to increased host quality (see Chapter 1). The lack of a significant relationship between Calluna nitrogen content and pupal weight (Fig. 5-8) is consistent with this hypothesis.
If O. brumata is insensitive to Calluna nutritional quality, then this contrasts strongly with the findings of previous studies on broadleaved hosts (e.g. Feeny, 1970). In order to resolve this apparent paradox, it is proposed here that O. brumata is more sensitive to changes in the quality of a high-quality host, such as oak, than of a poor-quality host, such as Calluna. A mechanism by which this could occur is discussed, with the aid of a simple conceptual model.
Nutritional compensation mechanisms (Section 1.4) are well-known to affect the apparent sensitivity of insect development to host nutrient content, and can operate to differing extents on diets of differing quality (Slansky & Feeny, 1977; Simpson & Simpson, 1990). Increased larval feeding in response to poor-quality diet can cause an apparent loss of developmental sensitivity to host nutrient status (Slansky & Feeny, 1977; Wheeler & Slansky, 1991), but this effect may be visible only above a certain host quality threshold (Ohmart et al., 1985). In order to explain the present results in such terms, it is proposed that O. brumata invokes compensation most strongly on a low-quality host such as Calluna, but can facultatively relax it on a better-quality plant. In this case, therefore, O. brumata invokes compensation below a quality threshold.
This hypothesis is illustrated schematically in Figure 5-9. In the absence of compensation mechanisms, the response of an insect herbivore to host nutrient status will be as shown in Figure 5-9i: developmental performance rises steadily with increasing host quality, until it reaches a "plateau" where host quality is no longer limiting (Thomas & Hodkinson, 1991). A critical minimum performance for herbivore viability (Pc) is associated with a critical minimum host quality (Qc), below which plants are unsuitable for herbivore development.
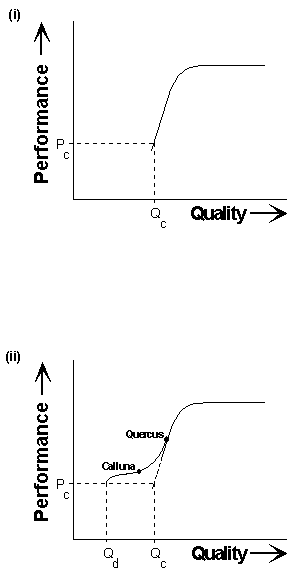
Fig. 5-9. Hypothetical model showing the effect of
nutritional compensation on herbivore response to diet quality;
(i): response without compensation; (ii): response with compensation, induced
progressively in response to declining host quality.
Compensatory feeding (Slansky & Feeny, 1977), induced progressively in response to low diet quality, will distort this response into a sigmoidal curve (Fig. 5-9ii). In this model, compensation is not invoked immediately diet quality begins to fall, since it is assumed that other costs associated with increased consumption (for example, increased risk of predation) must be "traded off" against the benefits of increased development. On a high-quality host such as Quercus, therefore, performance remains sensitive to changes in host quality (Fig. 5-9ii). However, below this quality threshold, compensation is induced and becomes progressively more important as food quality declines. At low host quality, compensation "flattens" the response of performance to quality, i.e. insect development appears insensitive to host quality, as on Calluna (Fig. 5-9ii). At a new, lower critical minimum host quality (Qd), compensatory feeding is no longer sufficient to supply the nutrients necessary to sustain growth, and performance then declines steeply.
This model predicts that O. brumata will show stronger compensatory feeding on Calluna than on Quercus. This remains to be tested experimentally, although Section 3.3.4 showed a high rate of frass production by O. brumata on Calluna, which is consistent with this prediction (see also Chapter 6).
A flexible feeding strategy, such as that described above, could be beneficial for a polyphagous herbivore such as O. brumata, enabling it to survive by compensation on a poor-quality host, whilst still retaining the ability to respond rapidly to a more favourable one. Operophtera brumata, therefore, conforms to the general description of an eruptive herbivore given by Price (1992), for which "the world really is green" due to their nutritional adaptability and consequent facility to utilise low-quality foliage (see Section 1.3.1). According to this view, factors other than diet quality operate at most times to maintain population densities below outbreak levels. This view is consistent with the ability of O. brumata to outbreak on an apparently low-quality host such as Calluna, and also with the lack of response of O. brumata to Calluna phenology shown in this study.
The difference in survival between oak and moorland O. brumata biotypes on Calluna (Fig. 5-6) is not attributable to their different hatching dates, since the imposed hatch delay had no significant effect on survival in either biotype. Moreover, this biotype effect was not mirrored in any other indicator of larval performance (Table 5-1). Although the difference in survival could have been caused by specialisation of the moorland biotype to feeding on Calluna (see Section 1.5), an alternative hypothesis is that the oak-feeding broods were somehow reduced in fitness. For example, reduced insect performance due to sub-lethal effects of vertically-transmitted viral disease has been implicated in the decline of forest Lepidoptera outbreaks (Regniere, 1984; Myers & Kuken, 1995; Rothman & Myers, 1996). The fact that many of the oak broods used here were obtained from the Bonar Bridge population at the peak of its outbreak density (Sections 2.3.2), and that this population declined sharply in the following year (J.E. Kerslake, pers. obs.), argues that this possibility cannot be discounted.
The moorland O. brumata biotype achieved close synchrony of hatch date with Calluna budburst, both in situ and in the undelayed treatment of the feeding trial (Figs 5-2 & 5-5). However, this synchrony is unlikely to be an adaptation of moorland populations to host phenology, since experimental hatch delay had no adverse effect on larval performance on Calluna. Other factors which could favour early hatching dates in moorland populations include avoidance of predators (Picozzi, 1981; Holliday, 1985) and parasitoids (Chapter 4), and the availability of Vaccinium myrtillus as an earlier-flushing alternative host (Chapter 3).
However, differences in hatching dates between populations may also be caused by local adaptation of egg development rates or thresholds to local climatic conditions (see Fraser, 1995). The differences observed here (Fig. 5-3) are consistent with this hypothesis, since the population from the exposed high-altitude Hunthill moorland hatched before those from the low-altitude Orkney site, under constant conditions (Fig. 5-3). Similar differences in hatching date between oak and spruce-feeding O. brumata populations were documented by Fraser (1995).
In summary, the hatching dates presented above show similar variability to the life history and metabolism traits investigated in Chapter 4: the observed moorland variations are more readily attributable to local climate than to specialisation on Calluna.
This study falsifies the first hypothesis given above (Section 5.1.1), that hatch-budburst synchrony is an important determinant of O. brumata performance on Calluna. There is, therefore, no evidence to support the second hypothesis (Section 5.1.1), that moorland O. brumata populations are adapted to hatching in synchrony with Calluna budburst. The variability in hatching date between populations can be explained as the result of metabolic adaptation to differing local climates. The evidence that moorland O. brumata populations perform better on Calluna than oak-feeding populations is inconclusive: although they show higher survivorship, there was no difference between biotypes in growth rate, development index or pupal weight.
The apparent unimportance of hatch-budburst synchrony in the O. brumata- C. vulgaris system is not surprising in view of the moorland environment. Scottish moorland covers a wide range of altitudes (Gimingham, 1972), and therefore represents a geographically extensive but climatically diverse and unpredictable resource for an invertebrate herbivore. Harsh climatic conditions on high-altitude moorlands can delay the onset of C. vulgaris growth in some years by more than two weeks (J.E. Kerslake, pers. obs.). High wind speeds in such sites are likely to cause extensive and unpredictable dispersal both of newly-hatched O. brumata larvae (Edlant, 1971), and of adult males. The evolution of a tight hatch-budburst synchrony would represent more of a disadvantage than an advantage in such a variable and dispersive system. In contrast, resilience both to variable host phenology and to low host quality are important characteristics enabling O. brumata to exploit this resource.
|
Previous Chapter |
 Contents Page |
Next Chapter |