|
Previous Chapter |
 Contents Page |
Next Chapter |
|
Previous Chapter |
 Contents Page |
Next Chapter |
Populations of O. brumata were obtained from low-altitude and high-altitude moorland outbreaks, and from an oak woodland. Their life histories, pupal respiration rates and parasitism rates were contrasted, and a reciprocal transfer experiment was conducted in order to test for nutritional specialisation of Calluna- and oak- feeding populations to their respective hosts. Differences in adult emergence date, respiration rate and parasitism were observed between populations, but there was no evidence for host specialisation. The observed inter-population differences are more likely to be due to altitude and climate than to differences in host quality.
Chapter 3 demonstrated that Calluna vulgaris is a sub-optimal host for O. brumata, and argued that the nutritional superiority of Vaccinium myrtillus provides only a partial explanation for the occurrence of moorland O. brumata outbreaks. This chapter considers four additional factors that may further increase the probability of these outbreaks: (i) specialised life history traits in moorland O. brumata populations; (ii) metabolic adaptation to the climatic conditions of Scottish moorlands; (iii) nutritional specialisation of moorland O. brumata populations to feeding on C. vulgaris; (iv) the availability of "enemy-free space" (Jeffries & Lawton, 1984). These factors were investigated by comparing the life histories, morphologies, growth rates on Quercus and Calluna, and parasitism levels of O. brumata from moorland and oak woodland populations.
The moorland climate varies greatly with altitude (Gimingham, 1972; Coulson & Whittaker, 1978), and moorland O. brumata outbreaks have occurred at altitudes from 50 m above sea level (on Orkney) to between 600 m and 650 m a.s.l. (on mainland Scotland) (see Chapter 2). This wide distribution presents an opportunity to distinguish the effects of altitude on O. brumata populations from the more general effects of the moorland habitat. In order to highlight altitudinal effects, O. brumata outbreak populations from two moorland types were studied: low-altitude Vaccinium-poor moorland on Orkney (Plate III) and high-altitude mixed Calluna-Vaccinium moorland on mainland Scotland (Plate I).
Operophtera brumata life history varies markedly between populations in different climates and habitats (Holliday, 1985). For example, the duration of the pupal stage is reduced at high latitude and high altitude (Schneider-Orelli, 1916; Uvarov, 1931, p.106), with the result that adult emergence dates vary from late September in Finland and northern Russia, to February in Sicily and Cyprus (Uvarov, 1931; Kozhanchikov, 1950). This variability was considered by Kozhanchikov (1950) to be caused by sensitivity of pupal development to ambient temperatures. However, other work has shown that pupal duration is insensitive to temperature over the range 9.5ºC to 16.8ºC (Holliday, 1983); furthermore, inter-population differences in pupal duration persist under constant conditions, and are heritable (Speyer, 1938; Wylie, 1960a). The latter result suggests that variability in this life history trait is sustained in the field by restricted gene flow between geographically separated populations.
This study tests the hypothesis that isolated moorland O. brumata populations exhibit specialised life history traits which enhance their survival or development in moorland systems. Selection pressures which could drive the evolution of such traits include climate (Section 3.4.3), synchronisation of the insect life cycle to patterns of food availability (Section 1.2.4), and avoidance of natural enemies (Holliday, 1985) (Section 1.3.3). Confirmation of the above hypothesis would provide evidence for restricted gene flow between moorland and non-moorland O. brumata populations, and any observed life history specialisation may also help to reveal the constraints and selection pressures acting on O. brumata in moorland systems.
In order to test the above hypothesis, the life histories of O. brumata from moorland and oak woodland outbreak populations were contrasted. The present study concentrates on the pupal stage and adult emergence, whilst Chapter 5 deals in detail with hatching phenology.
A common mechanism by which insects adapt their metabolism to cold climates is alteration of the relationship between respiration rate and temperature; this enables optimal respiration rate to be achieved at a lower temperature in an adapted population than in a non-adapted one (Block, 1990). Under uniform conditions, therefore, a cold-adapted insect will respire more rapidly than a non-adapted one. For example, increased pupal and larval respiration rates have been shown in a cold-adapted Alaskan population of Papilio canadensis (Lepidoptera: Papilionidae), compared with a population from Michigan, when both were reared at a uniform temperature (Kugal et al., 1991; Ayres & Scriber, 1994).
This study tests the hypothesis that different O. brumata populations show metabolic adaptation of this kind to their respective habitats. Cold-adaptation would be expected in populations which inhabit exposed high-altitude Scottish moorlands (Section 3.4.3). Confirmation of the above hypothesis would provide further evidence that O. brumata populations are adapted to their local habitats, and that limited migration and gene flow occurs between altitudes and habitat types.
Section 1.5 introduced the concept of "host races" (see also Fox & Morrow, 1981; Scriber, 1983), and the idea that specialisation to feeding on one host may demand a relative loss of performance on other host species (Rausher, 1984; Bernays & Graham, 1988). Evidence for such "trade-offs" (Section 1.5) has been found at the population level in a small number of cases (Rausher, 1982; Tabashnik, 1983; Nitao, 1991), although many other studies at the genotype level have failed to detect them (Via, 1984; Futuyma & Phillipi, 1987; Rausher, 1988).
This study tests the hypothesis that moorland outbreak O. brumata populations are specialised to feeding on C. vulgaris, and that improved performance on C. vulgaris is associated with a relative loss of performance on Quercus robur (pedunculate oak). Such specialisation may be necessary in order to deal with the high levels of secondary compounds present in the foliage of C. vulgaris (Iason et al., 1993).
In order to test the above hypothesis, a reciprocal transfer experiment (Via, 1990) was used to compare the growth rates of O. brumata larvae from moorland and oak woodland outbreak populations on both C. vulgaris and Q. robur.
Operophtera brumata populations in broadleaved woodlands are commonly subject to high levels of parasitism (Wylie, 1960b; Hassell, 1980), and parasitoids can exert a strong influence on O. brumata population dynamics (Embree, 1966; Roland, 1990, 1995). Section 1.3.3 introduced the hypothesis that exposed, high-altitude moorlands provide "enemy-free-space" for O. brumata (Gilbert & Singer, 1975; Jeffries & Lawton, 1984), and that the availability of this space is an important determinant of outbreak potential. This hypothesis was tested by assessing parasitism levels in moorland and oak woodland outbreak O. brumata populations.
Operophtera brumata larvae were collected from the Hunthill high-altitude moorland outbreak (Fig. 2-1ii) during June 1993, and from three outbreak sites during June 1994: Auchnafree high-altitude moorland in Perthshire (Fig. 2-2iii), Swartaback low-altitude moorland on Orkney (Fig. 2-1i), and Spinningdale oak woodland near Bonar Bridge (Fig. 2-2i). Table 4-1 summarises the characteristics of the three outbreak sites used in 1994, from which the majority of the data presented in this chapter were obtained.
Table 4-1. Characteristics of outbreak sites from which O. brumata larvae were sampled in 1994.
| Perthshire moorland | Orkney moorland | Bonar Bridge oak | |
| Site location | Auchnafree estate, Perthshire | Swartaback, mainland Orkney | Spinningdale near Bonar Bridge |
| altitude (a.s.l.) | 630 m | 30 m | 50 m |
| host-plants | C. vulgaris / V. myrtillus | C. vulgaris | Q. petraea |
| outbreak status | Medium larval density (200/m2). High densities in previous year. Exclusively O. brumata. | Consistent high densities (500/m2). Primarily O. brumata. | Extremely high densities in 1993 and 1994. Multi-species outbreak. |
| date sampled | 16-06-94 | 6-06-94 | 6-06-94 |
| number of larvae sampled | 71 | 228 | 125 |
| food plant used for rearing | V. myrtillus | C. vulgaris | Q. petraea |
Sampled larvae in their fourth or fifth instars were reared communally in 40 cm x 25 cm plastic jars, with a maximum of 50 larvae per jar. Separate jars were used for larvae from each sampled population, and a 5 cm layer of moist peat was supplied at the bottom of each jar. Larvae were reared on the host species from which they were originally taken (Table 4-1) until they either pupated or were killed by the emergence of parasitoids. Fresh foliage was supplied daily during the rearing period, and jars were stored at ambient temperatures in an unheated out-building near Aberdeen. Three weeks after pupation, parasitoid pupae and unparasitised O. brumata pupae were counted. Parasitism level was expressed for each population as the ratio of the number of parasitoid pupae to the total number of parasitoid and unparasitised O. brumata pupae present.
The unparasitised O. brumata pupae obtained in 1994 (Section 4.2.1) were weighed, sexed, placed into individual plastic test tubes, and covered with a 4 cm-deep layer of moist Irish moss peat mixed with an inert water-retaining polyacrylamide gel. The tubes were stored at ambient Aberdeen outdoor temperatures, and were monitored daily for adult emergence. A total of 311 pupae were monitored, from which 218 adults successfully emerged. Date of emergence was recorded for each adult, and fresh weight and male wing length (from the base to the distal tip of the forewing) were measured.
Twenty-five bare-rooted Quercus robur saplings, 6 years old and approximately 1.4 m in height, were obtained in March 1994 from a nursery in N.E. Scotland (Eastland Foresters, Mary Culter) and were established in 40 cm diameter pots filled with top-soil. Twenty-five turves of Calluna vulgaris, approximately 80 cm x 80 cm, were obtained from an upland moorland (O.S. NO 644802, altitude 450 m). Trees and turves were placed in an outdoor enclosure at I.T.E. Banchory (OS 676984, altitude 130 m) in early April 1994 and were watered daily. Wet moorland peat was packed around any exposed roots of the Calluna turves.
It was originally intended to rear moorland and oak-feeding O. brumata populations from eggs on these plants. Stocks of eggs were obtained for this purpose, and were placed into feeding bags which were tied onto the plants in May 1994. However, heavy mortality of O. brumata occurred during this attempted experiment, due to low egg viability and poor establishment of newly-hatched larvae. The latter problem may have been caused by a period of extremely hot and dry weather which coincided with larval emergence. Consequently, a second experiment was set up to measure the performance of larvae obtained from the field.
Stocks of O. brumata larvae were obtained on 6 June 1994 from Swartaback and from the Bonar Bridge oak outbreak, as described in Section 4.2.1. The Swartaback moorland outbreak was selected for use in this trial because the only host at this site was Calluna (Table 4-1), and larvae were therefore judged more likely to have become specialised to this host than those from a mixed Calluna-Vaccinium moor.
The feeding trial commenced on 13 June 1994, by which date the majority of Quercus buds were fully open, with individual leaves approximately 40 mm long and 30 mm wide, and new Calluna shoots were approximately 30 mm in length. Third-instar larvae were weighed and placed into individual Organza polyester feeding bags. A single bag was tied onto the shoots of each Quercus sapling or Calluna turf. The following permutations of O. brumata population and experimental food plant were set up:
Table 4-2. Design of reciprocal transfer feeding trial, 1994.
| Source population | Trial host plant | Number of replicates |
| Bonar Bridge (Quercus) | Quercus robur | 13 |
| Bonar Bridge (Quercus) | Calluna vulgaris | 13 |
| Orkney (Calluna) | Quercus robur | 12 |
| Orkney (Calluna) | Calluna vulgaris | 12 |
On 24 June 1994, after 11 days of feeding, surviving larvae were removed and re-weighed. Larvae were then replaced onto the same plants in order to complete their development to the pre-pupal stage. At this stage, larvae were removed from the plants and placed into 12 cm diameter plastic jars containing a layer of moist peat. Jars were stored at ambient outdoor temperatures for three weeks, after which they were searched for O. brumata or parasitoid pupae.
A total of 311 pupae were obtained from the larval rearing procedure: 71 from the Perthshire moorland population, 141 from the Orkney moorland, and 99 from the Bonar Bridge oak woodland. Significant differences in O. brumata pupal weights were observed between sexes, with males significantly heavier than females in both moorland populations (Perthshire: t69 = 3.26, p < 0.01; Orkney: t139 = 3.22, p < 0.01), but not in the Bonar Bridge oak woodland population (t97 = 0.97, n.s.) (Fig. 4-1). Pupal weights differed significantly between populations for both sexes (Fig. 4-1); this effect was due to the Orkney moorland population, reared on C. vulgaris, which attained significantly lower pupal weights than the other two populations (t126 = 2.57, p < 0.05 for males; t92 = 2.39, p < 0.05 for females).
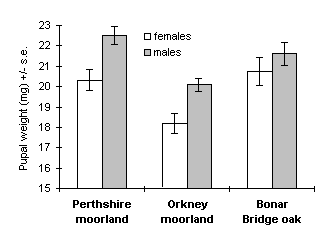
Figure 4-1. Pupal weights attained by O. brumata reared from
three source populations; effect of source population for females: F(2,141)
= 6.17, p < 0.01; males: F(2,164) = 8.11, p < 0.001
However, there was no significant difference between populations if the analysis was limited only to viable pupae (i.e. those from which adult moths subsequently emerged) (Fig. 4-2i), since a greater proportion of the lower-weight Orkney moorland pupae was non-viable (Fig. 4-2ii).
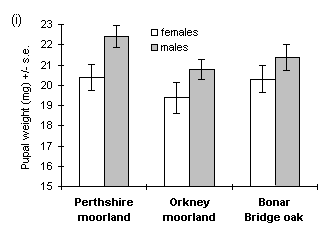
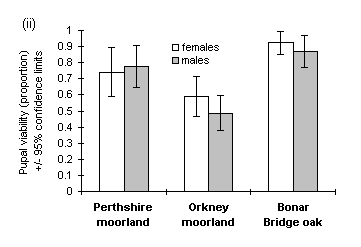
Figure 4-2. Pupal weights and viabilities of O. brumata reared
from three source populations. (i): Mean weights of only those pupae from
which viable adults emerged; effect of source population for females: F(2,105)
= 0.57, n.s.; males: F(2,107) = 1.95, n.s. (ii): Overall pupal viabilities,
with 95% confidence intervals for the binomial estimation.
Pupal viability was lower in males than in females (c2(1) = 4.89, p < 0.05), and declined significantly with decreasing pupal weight in males (rank correlation t = 0.714, p < 0.05 (Kendall, 1970)) but not in females (t = 0.429, n.s.) (Fig. 4-3). The lowest viable male pupal weight observed in this study was 12.4 mg, and the lowest viable female pupal weight was 10.2 mg (Fig. 4-3). This discrepancy between male and female viable pupal weights agrees with previous studies (Gradwell, 1974; Holliday, 1983).
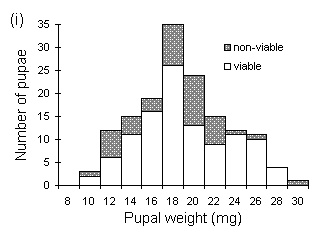
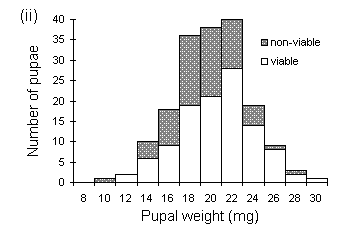
Figure 4-3. Distribution of weights of viable and non-viable
O. brumata pupae; (i): females; (ii): males..
Adult emergence of the Perthshire moorland population occurred considerably in advance of the emergences of the other two populations (Fig. 4-4). Peak rate of emergence of the Perthshire moorland population occurred between 3 and 5 November, whilst peak emergence rates of the Orkney moorland and oak woodland populations occurred more than three weeks later, between 30 November and 1 December (Fig. 4-4).
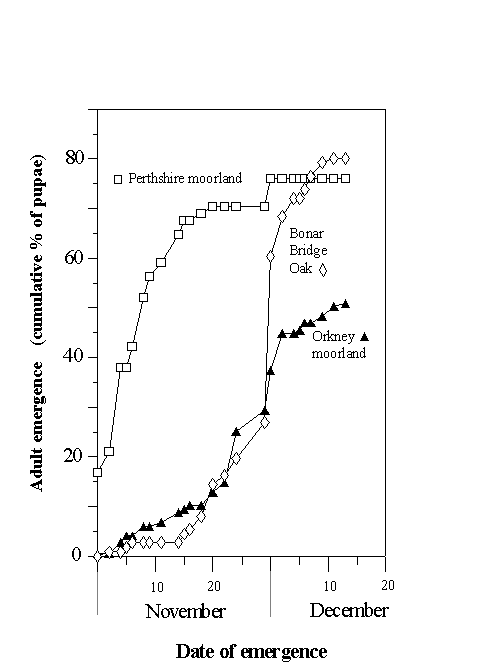
Figure 4-4. Dates of adult emergence in three populations of O.
brumata. Emergence is expressed for each population as the cumulative proportion
of the original stocks of pupae from which a live adult had emerged.
There was no significant relationship between pupal weight and date of adult emergence in either the Perthshire or the Bonar Bridge populations (Fig. 4-5). There was a significant negative relationship in Orkney moorland males, (Fig. 4-5iii), but the validity of this regression is questionable, since it is strongly influenced by a small number of outlying points.
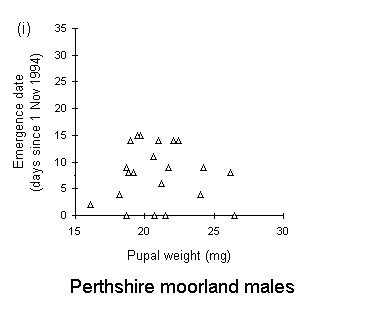
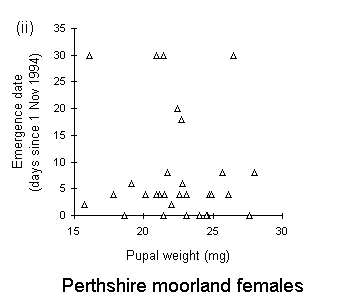
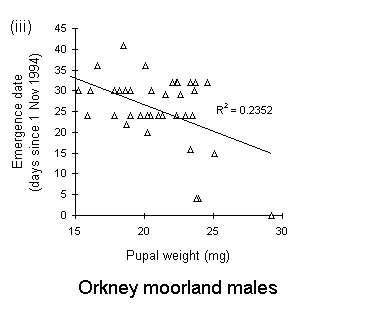
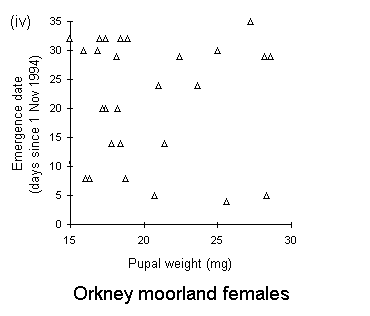

Figure 4-5. Relationships between early pupal fresh weight and timing of subsequent adult emergence for individual pupae from each O. brumata population and sex. A significant negative relationship is present in only one case: (iii) Orkney moorland males: F(1,37) = 11.4, p < 0.01, which is strongly influenced by a small number of outlying points.
The fresh weights of emergent adults differed markedly from the earlier pattern of pupal weights (Fig. 4-6 cf Fig. 4-2i). Adult females from all three populations showed almost identical fresh weights, whilst males from the two moorland populations were significantly lighter than those from the oak woodland population (t64 = 3.13, p < 0.01) (Fig. 4-6).
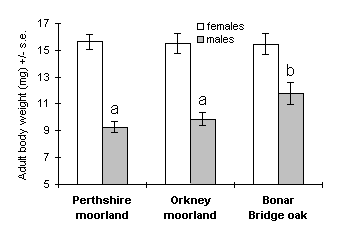
Figure 4-6. Mean adult fresh weights of O. brumata; effect of
source population for females: F(2,93) = 0.01, n.s.; males: F(2,63) = 5.14,
p < 0.01. For males, bars not sharing the same letter are significantly
different.
There was substantial weight loss between the early pupa and adult stages, which is attributable primarily to pupal respiration (Gradwell, 1974). Weight loss was considerably greater in males than in females (Fig. 4-7), and was significantly greater in males from the Perthshire moorland population than in males from the oak woodland population (t43 = 3.34, p < 0.01), with the Orkney moorland population intermediate between these values (Fig. 4-7).
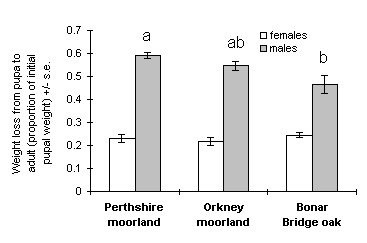
Figure 4-7. Proportion of weight lost between pupation and adult
emergence in O. brumata; effect of source population for females: F(2,93)
= 0.83, n.s.; males: F(2,63) = 6.66, p < 0.01. For males, bars not sharing
the same letter are significantly different.
Because pupal weight loss was more variable in males than in females (Fig. 4-7), pupal weight was a poorer predictor of adult fresh weight in males than in females (Fig. 4-8).
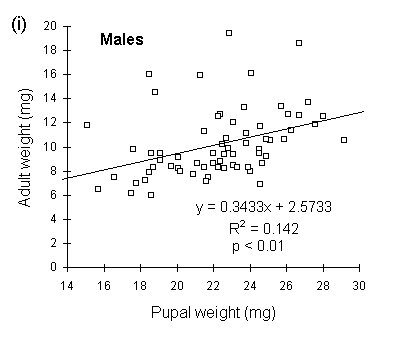
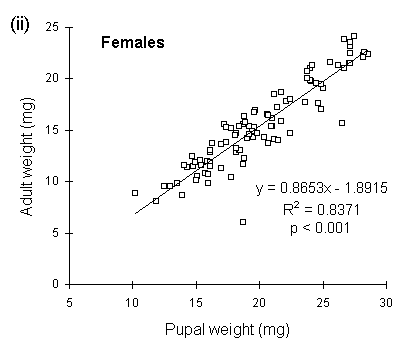
Figure 4-8. Relationship between pupal weight and subsequent
adult fresh weight for O. brumata (all three populations combined); (i):
males; (ii): females.
Pupal weight was a reasonably good predictor of adult male wing length (R2 = 0.324, p < 0.01). Male wing length was greater in the Perthshire and Bonar Bridge populations than in the Orkney population (Fig. 4-9i), and Perthshire males showed a significantly greater wing length to body weight ratio than the other populations (Fig. 4-9ii).
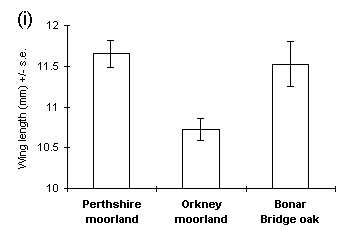
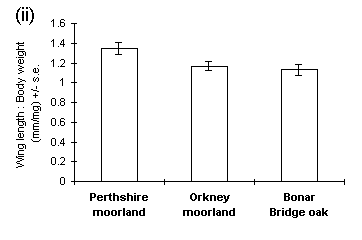
Figure 4-9. Male adult O. brumata morphology; (i) wing length;
effect of source population: F(2,43) = 6.0, p < 0.01; (ii) ratio of
wing length to body weight; effect of source population: F(2,42) = 4.33,
p < 0.05.
Larval growth data were analysed by two-way analysis of variance, with source population and trial food plant as factors. Specialisation of the Orkney moorland population to Calluna was expected to increase the growth rate of this population on Calluna, compared with the oak-feeding Bonar Bridge population on Calluna. A significant interaction between source population and trial host would provide evidence for a "trade-off" between performance on these hosts (see Section 4.1.4).
Data from 8 larvae were removed prior to analysis, since these larvae were found to have been parasitised. These parasitised larvae grew more slowly than unparasitised larvae (mean growth rate = 1.85 mg day-1 for parasitised larvae; 2.65 mg day-1 for unparasitised larvae; p < 0.05). A further 12 larvae died during the course of the feeding trial, leaving 30 final growth values for analysis. There was no evidence that larval growth over the experimental feeding period was proportional to initial weight (F(1,28) = 0.88, n.s.); larval weight gain over 11 days was therefore used as the response variable, rather than mean relative growth rate (see Appendix I).
There were significant effects both of source population and of trial host-plant on larval weight gain, although the former effect was weak (Table 4-3).
Table 4-3. Analysis of variance table for O. brumata weight gain in reciprocal transfer feeding experiment, 1994.
| Factor | d.f. | Mean Square | F-Ratio | p |
| Source population | 1 | 402.76 | 4.29 | 0.048 |
| Trial host-plant | 1 | 820.86 | 8.75 | 0.007 |
| Source x host interaction | 1 | 185.71 | 1.98 | 0.171 |
| Error | 26 | 93.79 | ||
| Total | 29 |
Mean growth of both populations was greater on Quercus than on Calluna (Fig. 4-10), and there was no evidence that larvae from the Orkney population grew more rapidly on Calluna than those from Bonar Bridge (Fig. 4-10). There was no significant interaction between source population and trial host (Table 4-3), and consequently no evidence for any "trade-off" between these hosts. However, the power of the analysisto detect such an effect was limited by the low numbers of surviving larvae, particularly in the Orkney population on Quercus (n = 5).
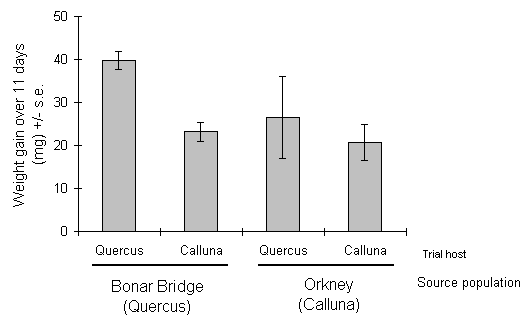
Figure 4-10. Fresh weight gain of O. brumata from Quercus and
Calluna- feeding source populations over 11 days in reciprocal transfer
feeding trial.
No parasitoids were found in either year of sampling in the high-altitude moorland O. brumata populations (n = 557 from Hunthill in 1993; n = 71 from Perthshire in 1994). No parasitoids were found by dissection of failed pupae from these populations.
In contrast, Phobocampe neglecta (Holmgren) (Hymenoptera: Ichneumonidae) was present at 27% in the low-altitude Orkney moorland population; Cyzenis albicans (Fallén) (Diptera: Tachinidae) and Lypha dubia (Fallén) (Diptera: Tachinidae) together achieved a 42% total parasitism rate in the Bonar Bridge population† (Fig. 4-11).

Figure 4-11. Parasitism levels (+/- 95% c.i.) in Orkney moorland
and Bonar Bridge oak woodland O. brumata populations.
Adult emergence in the Perthshire moorland O. brumata population occurred more than three weeks in advance of the emergence of Orkney moorland and oak woodland populations (Fig. 4-4). Emergence dates obtained in this rearing study are consistent with emergence dates in the field: peak adult abundance occurs in early to mid-November in high-altitude moorlands (J.E. Kerslake, pers. obs.) and in early December in oak woodland populations in Oxford (L. Cole, pers. comm.). There was little evidence for any relationship between pupal weight and adult emergence date (Fig. 4-5), and consequently little evidence to support the view of Kozhanchikov (1950) that larval foraging success is negatively related to the duration of the pupal stage. The variability in emergence dates between populations is, therefore, unlikely to be caused by the differing nutritional qualities of the hosts on which they were reared (Table 4-1).
Early adult emergence at the high-altitude Perthshire site is probably an adaptation to climate, and is not an adaptation to feeding in moorland systems generally, since the low-altitude Orkney moorland population emerged at the same time as the woodland population (Fig. 4-4). These results agree with the work of Schneider-Orelli (1916), who found that O. brumata adult emergence dates were earlier in high-altitude populations in the Alps than in neighbouring low-altitude populations. Holliday (1985) speculated that harsh climatic conditions are an important constraint on O. brumata life history, and adult emergence has been shown to be disrupted by snow cover (see Section 3.4.3). It is therefore probable that early emergence in high-altitude moorlands is the result of selection pressure to complete mating and oviposition before the arrival of the heavy snowfalls, which are characteristic of such sites. The present results, therefore, support the hypothesis that moorland O. brumata populations have evolved life history adaptations which enhance their survival in this habitat, and reinforce the view that patterns of early winter snowfall have a strong impact on O. brumata population density.
Selection for early adult emergence may, however, be countered by other advantages of late emergence, such as a reduced risk of predation (Holliday, 1985). It is probable that the divergent times of adult emergence observed in different populations represent the outcomes of trade-offs between such risks, which vary with habitat type and altitude.
Significant differences were observed between populations in male weight loss between the early pupa and adult stages (Fig. 4-7). Pupal weight loss is the result of respiration, and is always higher in males than in females of O. brumata due to the more profound metamorphosis undergone by males (Gradwell, 1974). The requirement for a high respiration rate in male pupae accounts for the positive relationship between early pupal weight and pupal viability shown for males in this study (Section 4.3.1) (see also Holliday, 1983).
The results shown in Figure 4-7 imply that male pupal respiration rate was higher in the high-altitude moorland O. brumata population than in the oak woodland population under identical temperature regimes. As discussed above (Section 4.1.3), such a difference in respiration rates under uniform conditions suggests that metabolism in the high-altitude population is adapted to cold temperatures. Metabolic adaptation may enable O. brumata populations to achieve optimal respiration rates at the ambient temperatures specific to their different habitats.
An alternative interpretation of the observed difference in pupal weight loss is that male O. brumata respiration rate is increased on high-altitude moorlands for some other reason than ambient temperature. For example, the advanced date of adult emergence in the high-altitude population (Section 4.4.1) may demand an accelerated rate of metamorphosis. Studies of respiration rates and temperature regimes in the field would be necessary in order to distinguish between the above alternative hypotheses, and to determine whether O. brumata populations are indeed capable of achieving similar respiration rates in such different climates and habitats.
The interpretation of the reciprocal transfer experiment (Section 4.3.4) is limited both by the relatively low level of replication and by the fact that the larvae used were obtained directly from the field, with associated problems of parasitism and possible habituation to their indigenous host-plants. Furthermore, the use of late-instar growth rate as the sole measurement of O. brumata performance in this experiment is restrictive. Under more favourable circumstances, pupal weight would also have been measured, but in the present experiment insufficient numbers of O. brumata survived to the pupal stage to give meaningful data.
Given these constraints, the results presented here fail to support the hypothesis that the moorland O. brumata population tested was specialised to feeding on Calluna. The growth rate of the moorland population on Calluna was not significantly different from that of the oak-feeding population (Fig. 4-10), and there was no significant difference between populations in their relative performances on Calluna and Quercus (Table 4-3). Although the oak-feeding population grew particularly rapidly on Quercus, there was clearly no major barrier to growth of this population on Calluna, or of the moorland population on Quercus.
Whilst Sections 4.4.1 and 4.4.2 argue for the existence of life history and metabolic adaptations in moorland O. brumata populations, the present null result argues against the formation of distinct "host races" (Section 1.5) on Calluna. However, this issue is addressed again in Chapter 5, where the performance of O. brumata from different source populations on Calluna is investigated in more detail.
The lack of detectable parasitism in the two high-altitude moorland O. brumata populations contrasts sharply with the high levels found in both the low-altitude moorland population on Orkney and the oak woodland population at Bonar Bridge (Fig. 4-11). This result is similar to previous work by Jordan (1962), who found that parasitism levels in the moth Coleophora alticolella Zell. declined at high altitudes on moorland.
In attempting to explain the apparent low levels of parasitism on high-altitude moorlands, Coulson and Whittaker (1978) proposed that hunting by parasitoids is inhibited at such sites, due to harsh climatic conditions. An alternative explanation in this case is that the recent occurrence of O. brumata outbreaks on these mainland sites has preceded the ability of parasitoids to colonise them. In either case, the opportunity to escape from parasitism on high-altitude moorlands, either spatially or temporally, may increase the probability of O. brumata outbreaks. The fact that the majority of moorland O. brumata outbreaks in mainland Scotland have occurred at altitudes above 500 m supports this hypothesis (see Chapter 8 for further discussion).
Significant differences were observed between populations in male wing length (Fig. 4-9i), and these differences corresponded more closely to the pattern of early pupal weights (Figs 4-1 & 4-2i) than to the body weights of emergent adults (Fig. 4-6). This suggests that wing length is determined at an early stage of pupal development, prior to the variable effects of pupal respiration on body weight (Fig. 4-7). If this hypothesis is correct, then the relationship between male wing length and body weight (Fig. 4-9ii) may be a purely passive function of initial pupal weight and subsequent pupal respiration. For this reason, it could be misleading to view variability in "wing loading" between populations as an adaptive response to their differing dispersal requirements (Ruohomäki, 1992).
Field-sampled larvae were reared communally at the start of this study (Section 4.2.1), an approach which was dictated by available time and labour. The subsequent statistical analyses regarded individual insects as independent units (Section 4.3), and are open to criticism on the grounds of pseudoreplication (Hurlbert, 1984). Whilst individual O. brumata were stored separately from the early pupal stage until adult emergence, it is nevertheless possible that between-jar variability in rearing conditions contributed to the subsequent inter-population differences in life history, metabolism and morphology shown above (Section 4.3). More rigorous and labour-intensive sampling in future studies may be able to clarify this point, but in the present case the above inter-population comparisons should be interpreted in the light of this criticism.
This study has provided evidence for adaptations of O. brumata life history and metabolism to the moorland environment, but no evidence of nutritional specialisation to Calluna, the dominant moorland host. The observed variations in O. brumata life history and metabolism are consistent with previous studies of Lepidoptera in other habitats (Schneider-Orelli, 1916; Kugal et al., 1991), and appear to be more closely related to altitude and climate than to host quality. Feeding on Calluna per se is unlikely to be responsible for the observed variability in life history and metabolism, because the low-altitude Orkney moorland population showed characteristics which were either similar to the oak woodland population (Fig. 4-4), or intermediate between the oak and the high-altitude moorland populations (Fig. 4-7).
Nevertheless, this study demonstrates that O. brumata life history and physiology are flexible, and suggests that high-altitude moorland populations are reproductively isolated from those at lower altitudes. The high-altitude climate imposes an increased risk of disruption of O. brumata adult emergence (Section 3.4.3), and necessitates changes in phenology and metabolism (Sections 4.4.1 & 4.4.2); however, this constraint is evidently balanced by the ability to escape from parasitism (Section 4.4.4). The higher parasitism rate experienced by O. brumata on Orkney (Fig. 4-11) reinforces the view of Section 3.4.3, that populations on such low-altitude moorlands will show different dynamics from those on higher-altitude moorlands (see Chapter 8 for further discussion).
† Parasitoids were identified by Dr. M. Shaw, Royal Museum of Scotland, Edinburgh.
|
Previous Chapter |
 Contents Page |
Next Chapter |