|
Previous Chapter |
 Contents Page |
Next Chapter |
|
Previous Chapter |
 Contents Page |
Next Chapter |
Feeding trials were used to compare O. brumata performance on Calluna vulgaris with that on Vaccinium myrtillus, which is an alternative host available on some moorlands. Vaccinium was clearly a better host than Calluna, and supported the establishment of newly-hatched larvae prior to Calluna budburst. The hypothesis that Vaccinium enhances O. brumata performance and increases the probability of outbreaks is discussed. Population data shows that release from the climatic constraint of early winter snowfall is another potentially important determinant of outbreak potential at high altitudes. However, neither of these factors can apply on low-altitude Orkney moorlands, where O. brumata populations may be expected to exhibit differing dynamics from those on higher-altitude mainland sites.
The recent outbreaks of Operophtera brumata on moorlands in Scotland are surprising because the dominant moorland host, Calluna vulgaris, is low in nitrogen and high in secondary compounds and fibre (Iason et al., 1993), and is therefore likely to be of poor nutritional quality (Section 2.2.2). This chapter tests the hypothesis that Calluna is a sub-optimal host for O. brumata, and examines the rôle of an alternative host species in supporting moorland O. brumata populations. Monitoring data of moorland O. brumata populations obtained between 1992 and 1994 are presented, and a "climatic release" hypothesis to explain the observed patterns of population change is presented and discussed.
Chapter 1 has shown that host-plant nutritional quality is a central issue in insect ecology, and that its rôle in affecting phytophagous insect abundance and development is sometimes contentious. One key objective of the present study is to determine to what extent O. brumata populations are limited by the presumed poor quality of C. vulgaris. Evidence that the "bottom-up" effect of host quality is a major constraint on O. brumata would give support to the hypothesis introduced in Section 1.2, that the current moorland outbreaks are caused by nutrient enrichment or "stress" of moorland vegetation. Failure to demonstrate such a constraint would support the view given in Section 1.3, that O. brumata is highly nutritionally flexible, and more likely to be limited by "top-down" or abiotic factors (Price, 1992; Hunter & Price, 1992).
Operophtera brumata development has been shown to be sensitive to the quality of some hosts: Feeny (1970) found that O. brumata development on oak was inhibited by leaf toughness and tannin content, and Wint (1983b) found that levels of available leaf protein in a range of broadleaved hosts correlated significantly with indices of larval performance. However, at the population level, Hunter et al. (1991) found no relationship between the occurrence of O. brumata outbreaks on Sitka spruce and the nutrient status of affected trees.
One factor which may counteract the presumed poor quality of C. vulgaris is the availability on some moorlands of Vaccinium myrtillus (bilberry) as an alternative host. Vaccinium myrtillus could be a superior host to C. vulgaris, because it has higher foliar nitrogen concentrations than C. vulgaris during the O. brumata feeding period (Powell & Malcolm, 1974; Lähdesmäki et al., 1990). If this is the case, then the presence of Vaccinium†, in addition to Calluna, on a moorland will increase the overall quality of the food resource available to O. brumata, and this may increase the probability of outbreak.
In order to test this hypothesis, patterns of O. brumata host exploitation were studied in two moorland outbreaks where both Calluna and Vaccinium were present. Larval distribution and development on Calluna and Vaccinium were investigated in the field, and development on these hosts was also measured in feeding trials.
Long-term population studies of O. brumata in woodland systems have contributed greatly to the development of life table analysis techniques (Varley & Gradwell, 1968), and to an understanding of the rôle of density-dependent mortality factors in regulating populations (Roland, 1990, 1994). In the present study, such long-term population monitoring was not possible. However, the dynamics of outbreaking moorland O. brumata populations are of fundamental importance, since no previous work has established whether densities remain stably high between years, or alternatively rise and decline rapidly. Furthermore, correlations between population changes and variable aspects of the moorland environment may suggest mechanisms by which outbreaks occur.
Section 1.3.2 reviewed the somewhat mixed evidence that "climatic release" can be a cause of insect outbreaks. Climatic constraints could be important for moorland O. brumata populations, since exposed moorlands have highly variable climates, particularly at high altitudes (Gimingham, 1972; Watson et al., 1994). In particular, the long-lasting snow cover which is common on high-altitude moorlands (Watson et al., 1994) may hamper adult emergence (Cuming, 1961). Occasional "release" from this constraint in some years could create the potential for outbreak. In order to test this hypothesis, O. brumata populations were monitored in a high-altitude moorland outbreak in mainland Scotland, and also in a low-altitude moorland outbreak on Orkney. The observed population changes are discussed in relation to the differing patterns of early-winter snow cover at these sites.
The distribution of newly-emerged O. brumata was investigated at a moorland outbreak site in Glen Shee (O.S. NO 142765, altitude 630 m) (Fig. 2-2ii), where vegetation was dominated by patches of C. vulgaris and V. myrtillus. Larval population densities were assessed on five dates between 8 April and 24 May 1993. On each occasion, seven samples of V. myrtillus and seven samples of C. vulgaris were taken from randomly-selected positions within the outbreak site. Samples were obtained by placing a 20 cm x 20 cm quadrat over a patch of the selected plant species. All vegetation of that species within the quadrat was then cut, placed into a polythene bag and removed to the laboratory. Each sample was then thoroughly searched for O. brumata larvae. In the case of Calluna samples, this simply involved an external examination of the foliage and stems. However, for Vaccinium samples, each leaf bud was given a score according to its stage of development (Fig. 3-1) and was then dissected in order to search for any larvae which had burrowed into it. A total of 3362 Vaccinium leaf buds were dissected in this way during the course of the study.
All larvae found were reared in jars on excised Vaccinium foliage until their final instars in order to facilitate species identification.
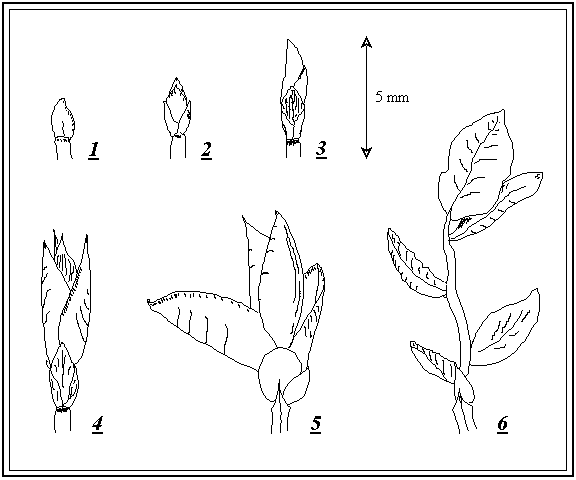
Figure 3-1. Arbitrary scoring system used to describe the stage of development of Vaccinium myrtillus leaf buds.
1: Bud fully closed; 2: Bud scales split; 3: Roll of leaves protruding from bud; 4: Individual leaves visible; 5: Individual leaves separated; 6: Stem extended.
Operophtera brumata distribution was assessed late in the feeding season at an outbreak near Black Hill, part of the Hunthill estate, in Tayside Region (O.S. NO 464755, altitude 640 m) (Fig. 2-1ii). Estate workers first reported O. brumata infestation at this site in 1991, and subsequently observed an increase in the affected area during 1992. The dominant vegetation at this site consisted of a mosaic of partially-defoliated C. vulgaris and V. myrtillus.
Larvae were sampled on 14, 23 and 30 June 1993. Samples were obtained by placing a 20 cm x 20 cm quadrat over selected categories of vegetation within the outbreak area. The following categories of vegetation were designated:
(i) areas of Calluna - dominated vegetation ( > 90% ground cover C. vulgaris);
(ii) areas of Vaccinium - dominated vegetation ( > 90% ground cover V. myrtillus);
(iii) areas of intermixed Calluna and Vaccinium, with neither species dominant.
Within each vegetation category, quadrats were placed randomly. Eight quadrat samples were taken from each of categories (i) and (ii) on each sampling date. Sixteen samples were taken from category (iii) on each occasion: eight from patches of Calluna within mixed areas, and eight from patches of Vaccinium. All vegetation within each quadrat was removed to the laboratory, where all larvae present were counted and classified into instars according to head capsule width, using the measurements given by Cuming (1961). An index of larval development was calculated for each quadrat sample as follows:
Development index = ![]()
where i = instar stage and pi = proportion of larvae in instar i.
Larvae collected by this procedure were reared to pupation in 30 cm-diameter plastic jars which were stored outdoors at I.T.E. Banchory (O.S. NO 676984, altitude 130 m a.s.l.) at ambient temperatures. Larvae were reared on the host species from which they were originally sampled. Host foliage was excised from turves obtained from the study site. Pupal weights were determined 14 days after the pre-pupal larvae had burrowed into moist peat which was provided in the rearing jars. Pupae were kept in moist peat at ambient temperatures until adult emergence. Fresh weights and body lengths (including head but excluding antennae) of adult moths were measured within 24 hours of emergence. Wing lengths of male moths were measured from the base to the distal tip of the forewing.
Adult O. brumata were obtained from the Hunthill outbreak site (Fig. 2-1ii) during November 1992 and maintained as a laboratory stock. Eggs from this stock were held at between 1º and 2º C until late April 1993, when they were transferred to ambient temperatures.
Ten turves of V. myrtillus (approx. 70 cm x 70 cm) and ten individual plants of C. vulgaris (approx. 70 cm in diameter) were obtained from an upland moorland (O.S. NO 644802, alt. 450 m a.s.l.). Both sets of plants were placed in an outdoor enclosure at Banchory, and wet peat was packed around any exposed roots.
Larval feeding bags of size 10 cm x 20 cm were constructed from "Organza" fine-mesh polyester. Five O. brumata eggs were placed into each bag, and two bags were tied onto the shoots of each individual plant or turf. This experiment was initiated on 10 May 1993, using eggs whose blue colour indicated that hatching was imminent. Bags were inspected regularly to ensure that the available food material within them had not been exhausted by the feeding larvae, although this problem did not occur. After 25 days, all bags were removed, surviving larvae counted and their developmental stages determined.
Larvae from the Hunthill laboratory stock described above were reared communally on excised foliage of V. myrtillus until the third instar. Attempts to rear a second stock of larvae on excised C. vulgaris foliage failed due to high mortality on this host. At the end of the third instar, larvae were transferred to individual 8 cm-diameter plastic pots. Feeding trials commenced at the start of the fourth instar in late May, and continued until the pre-pupal stage. Twelve larvae were fed on cut sprigs of V. myrtillus, and twelve on cut sprigs of C. vulgaris. Plant material was cut from turves obtained from an upland moor (O.S. NO 644802 , alt. 450 m a.s.l.). All sprigs of C. vulgaris had at least 2 cm of current season's growth at the start of the experiment. Fresh plant material was substituted every two days. Pots were stored in a shaded outdoor location at Banchory. The mean daily maximum ambient temperature over this period was 14.8º C, and the mean daily minimum was 8.2º C. Frass was collected every two days, and its fresh weight was determined. Weight of food eaten was estimated from the difference in fresh weight of the plant sprigs before and after larval feeding. Control pots without larvae were used to determine the contribution of plant water loss to this weight change, and a correction for this factor was applied by means of the following formula (from Waldbauer, 1968):
Corrected weight of food eaten = [ 1 - a/2 ] [ W - ( L + bL) ]
where W = weight of food provided;
L = weight of uneaten food;
a = ratio of water loss of control foliage to its initial weight
b = ratio of water loss of control foliage to its final weight
A humid atmosphere was maintained inside the pots by means of damp filter paper, in order to minimise plant water loss. However this factor remained a major source of error in these trials, and consequently precluded the calculation of reliable nutritional indices (Waldbauer, 1968). After feeding, pre-pupal larvae were weighed and allowed to burrow into moist peat. Growth rate was calculated as fresh weight gain over the course of the feeding trial divided by total feeding time (see Appendix I). Pupae were weighed 14 days after pre-pupal burrowing.
Adult O. brumata densities were monitored at Hunthill (Fig. 2-1ii) during the winters of 1992 and 1993, using sticky traps. Each trap was constructed from a 20 cm-wide strip of polythene coated with "Oecotac" (Oecos Ltd., Kimpton, Herts.) adhesive, wrapped around a 1.5 m tall, 8 cm square cross-section wooden fencing post at a height of 40 cm from the ground, and secured with drawing pins (Fig. 3-2). Adult O. brumata females show a strong climbing behaviour (Graf et al., 1995), and are readily trapped as they climb these posts. Males are more likely to be trapped by accidental contact with the adhesive surface during flight (J.E. Kerslake, pers. obs.).
Figure 3-2. Design of sticky trap for monitoring adult O. brumata population densities (see also Plate II).
Arrays of five fencing posts were set out in three regions of the Hunthill outbreak site: (i) at the centre of the area of defoliated Calluna; (ii) at the periphery of the defoliated area; and (iii) at a distance of 20 m beyond the visibly defoliated vegetation, in adjacent undamaged Calluna (Fig. 3-3). Each set of five posts was arranged in a cross formation (Fig. 3-3), with the perimeter posts 10 m from the centre post, and sticky bands were affixed to each of the four perimeter posts of each block. The centre posts initially carried pheromone traps, but these traps proved ineffective, possibly due to the high wind-speeds at this exposed site.
Sticky traps were in place at Hunthill by 12 November 1992, and by 20 October 1993. After these dates, all traps were checked once every week until either the emergence of adult O. brumata ceased, or the site became inaccessible due to snow cover. Numbers of additional O. brumata trapped since the previous week were recorded. Traps were re-set with fresh sticky bands every second week.
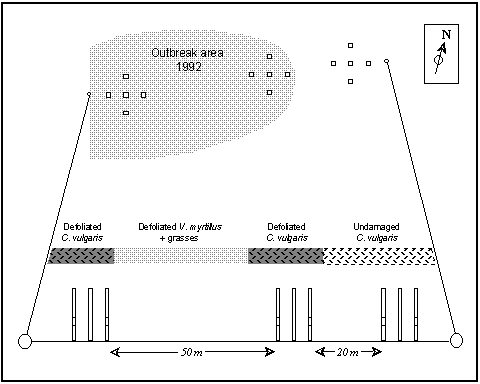
Figure 3-3. Positions of fence posts (with attached sticky traps) and vegetation types at the Hunthill outbreak site, November 1992.
Assessments of larval density were made at Hunthill in June 1993, and again on 16 June 1994. Larval densities were determined by examination of cut vegetation from a 20 cm x 20 cm quadrat, as described above (Section 3.2.1). The sampling strategy in 1993 was described above (Section 3.2.1). In 1994, six quadrat samples of Calluna and five samples of Vaccinium were taken from mixed Calluna-Vaccinium stands.
On Orkney, larval density was measured in a moorland outbreak at Swartaback (Fig. 2-1i), where O. brumata infestation was first noted in 1992 (A. MacDonald, pers. comm.). Vegetation was dominated by tall stands of Calluna, with no Vaccinium present. Twelve 20 cm x 20 cm quadrat samples were taken on 6 June 1994, and six on 6 June 1995.
No Operophtera brumata larvae were found on Calluna vulgaris during the course of the early-season study at Glen Shee, despite thorough searching of samples. No new season's growth was observed on any of the sampled Calluna shoots until 25 May, when new shoots were approximately 1 mm in length (J.E. Kerslake, pers. obs.)
In contrast, larvae were frequently found feeding inside the developing buds of Vaccinium myrtillus, where they spun the leaves together with silk. There was a close temporal relationship between the opening of V. myrtillus buds and their occupancy by O. brumata larvae (Fig. 3-4), and by 4 May, 17% of all examined buds contained a feeding larva. Small entrance holes were often observed on the outside of the developing leaf buds, and frass was frequently observed inside the buds.
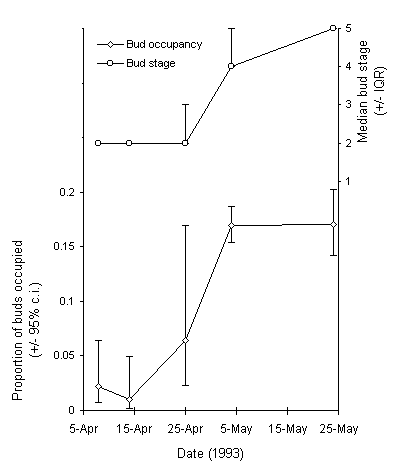
Figure 3-4. Development of the buds of Vaccinium myrtillus, and occupancy of those buds by feeding O. brumata larvae at Glen Shee outbreak, spring 1993. Effect of sampling date on occupancy (logit link model) F(1,47) = 5.14, p < 0.05.
Larval densities were extremely high at the Hunthill moorland outbreak site during June 1993 (Fig. 3-5), and severe defoliation of both C. vulgaris and V. myrtillus was observed during the course of the study. The highest larval densities were recorded on the first sampling date on C. vulgaris growing in "mixed" Calluna - Vaccinium stand types (Fig. 3-5). Densities were generally higher on Calluna than on Vaccinium. Within areas of mixed vegetation, larval densities were significantly higher on Calluna than on Vaccinium on all three sampling occasions (Mann-Whitney U-tests, p < 0.05 for each date).
Mixed stand types initially supported greater larval densities than the corresponding areas of "pure" vegetation dominated by one or other host. However, significant changes in larval density occurred between sampling dates in all four vegetation categories (Kruskal-Wallis tests, p < 0.01 for all categories); densities declined with time in areas of mixed vegetation and in areas of pure Vaccinium, but increased between 14 and 23 June in the pure Calluna vegetation category (Fig. 3-5).
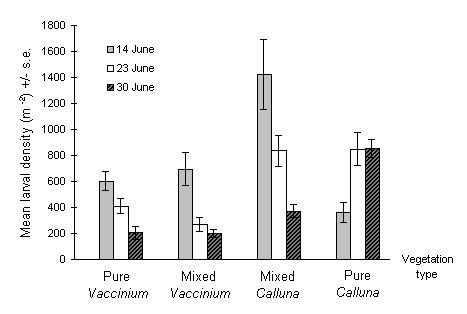
Figure 3-5. Mean larval densities of O. brumata in pure and mixed stands of Calluna vulgaris and Vaccinium myrtillus at Hunthill moorland outbreak site on three sampling dates in 1993.
Development indices were significantly lower in larval samples taken from areas of pure Calluna than from the other three vegetation categories on 14 and 30 June (Mann-Whitney U-tests, p < 0.01, p = 0.01 respectively) (Fig. 3-6). No other significant differences in development were observed in field-sampled larvae.
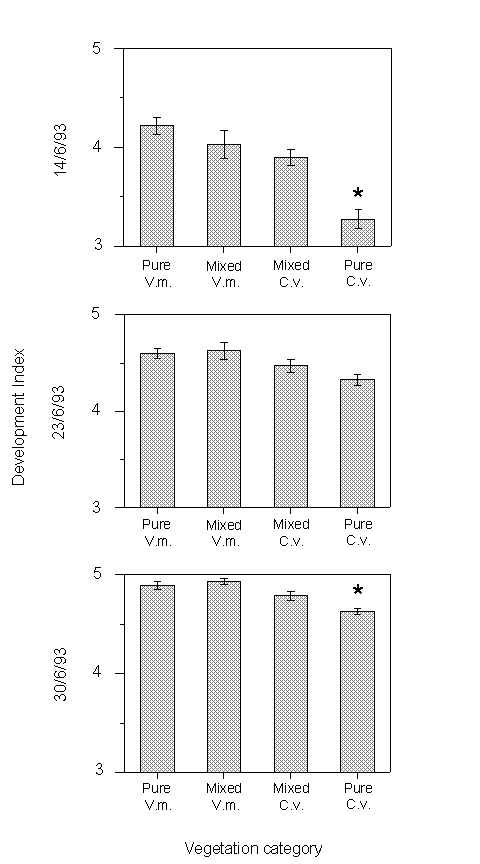
Figure 3-6. Mean larval development index (± s.e.)
in pure and mixed stands of Calluna vulgaris (C.v.) and Vaccinium myrtillus
(V.m.) on three sampling dates at Hunthill moorland outbreak site. Development
index was significantly lower in pure stands of Calluna than in the other
vegetation classes on two occasions (indicated by *).
Rearing of field-sampled larvae resulted in a total of 557 pupae, 76.7% of which produced viable adults. There was no significant effect of host species on pupal viability (c2(1) = 3.19, n.s.). The sex ratio of emergent adults was 1.1:1 male:female. Sex ratios showed no significant differences between host species, stand types (i.e. "pure" versus "mixed) or sampling dates (c2(1) = 0.482, 0.154, 0.365 respectively, n.s. for all tests).
To prevent pseudoreplication (Hurlbert, 1984), analysis of variance was performed on the mean pupal weight values from each combination of vegetation category and sampling date. The same approach was taken with adult fresh weight and size data. Significant effects of host species were observed on pupal weight (Fig. 3-7), male adult weight and male adult body length (Table 3-1). In each case, mean values were greater for larvae feeding on Vaccinium than on Calluna. Male wing length, female body length and female adult weight showed no significant effects of host species, although their mean values were consistently higher on Vaccinium than on Calluna (Table 3-1). Sampling date and stand type showed no significant effects in any of the analyses.
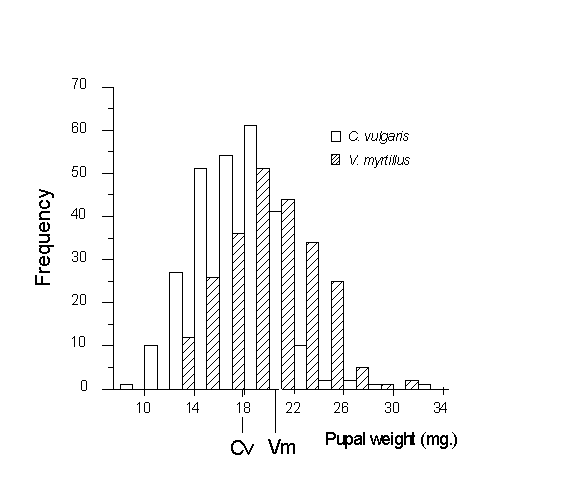
Figure 3-7. Distribution of pupal weights attained by field-sampled
O. brumata larvae on Calluna vulgaris and Vaccinium myrtillus (effect of
host species: F(1,6) = 19.11, p < 0.01). Mean weights on C. vulgaris
(Cv) and V. myrtillus (Vm) are indicated.
Table 3-1. Effects of host species on subsequent adult morphology in field-sampled O. brumata (ANOVA was performed on sample means from each combination of sampling date and vegetation category).
| Parameter | Mean (s.e.) for
C. vulgaris |
Mean (s.e.) for
V. myrtillus |
F - value
(d.o.f.) |
p |
| Female body weight (mg) | 11.99 (0.400) | 13.65 (0.584) | 3.14 (1, 5) | n.s. |
| Male body weight (mg) | 7.69 (0.248) | 9.02 (0.431) | 13.66 (1, 5) | < 0.05 |
| Male wing length (mm) | 9.89 (0.072) | 10.38 (0.103) | 6.13 (1, 5) | n.s. |
| Female body length (mm) | 6.27 (0.061) | 6.49 (0.080) | 1.64 (1, 6) | n.s. |
| Male body length (mm) | 7.28 (0.061) | 7.78 (0.075) | 14.78 (1, 5) | < 0.05 |
The eggs used in this feeding trial hatched concurrently on both hosts, and consequently both sets of larvae experienced an equal potential feeding time of 25 days. Larval survival after 25 days was significantly higher on Vaccinium than on Calluna (Fig. 3-8i). Larval development indices, calculated separately for each plant or turf, were significantly greater on Vaccinium than Calluna (Fig. 3-8ii).
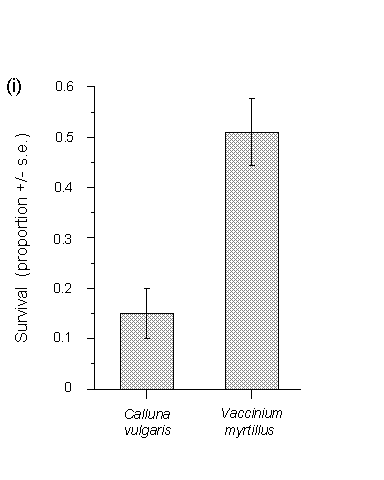
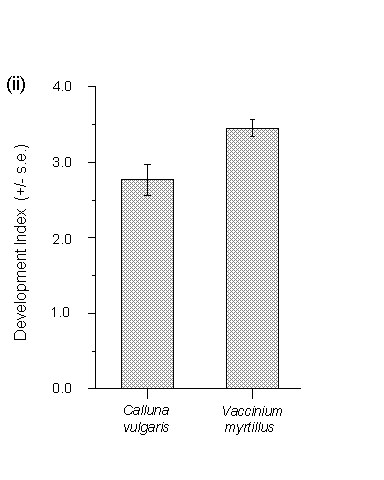
Figure 3-8. Performance of O. brumata larvae in feeding bags
after 25 days on Calluna vulgaris and Vaccinium myrtillus; (i): mean survival;
effect of host species (logit link analysis) F(1,18) = 30.54, p
< 0.01; (ii): mean larval development index; effect of host species
(Mann-Whitney U-test) p < 0.05.
Larvae reared on Calluna produced large amounts of frass, and the ratio of frass production to larval biomass gain was significantly greater on Calluna than on Vaccinium (Fig. 3-9i). The ratio of frass produced to food eaten appeared to exceed unity on this food plant (Fig. 3-9ii), an anomaly which was probably due to errors in the measurement of the weight of ingested food due to plant water loss (discussed above, Section 3.2.4). Larval growth rate was significantly higher on Vaccinium than on Calluna (Fig. 3-9iii), although there was no significant effect of host species on larval development time (c. 18 days to pre-pupal stage; Mann-Whitney U-test, n.s.). Pupal weight was significantly higher on Vaccinium than on Calluna (Fig. 3-9iv).
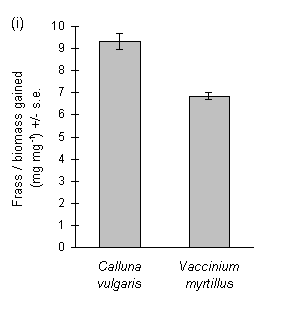
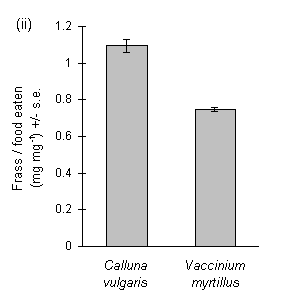
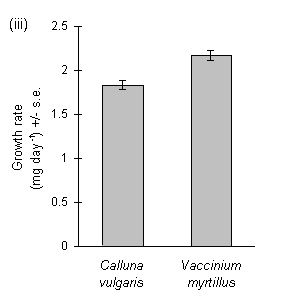
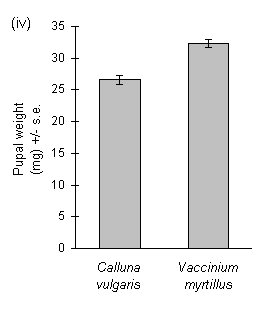
Figure 3-9. Performance of O. brumata larvae in feeding trials
on Calluna vulgaris and Vaccinium myrtillus; (i): ratio of fresh weight
of frass produced to larval biomass gained; effect of host species: t19
= 6.16, p < 0.01; (ii): ratio of fresh weight of frass produced to corrected
fresh weight of food ingested during trial; effect of host species: t19
= 9.48, p < 0.01; (iii): larval growth rate (calculated from untransformed
fresh weight gain); effect of host species: t19 = 4.29, p < 0.01;
(iv): pupal fresh weight; effect of host species: t17 = 5.82, p
< 0.01.
The numbers of adult female O. brumata trapped at Hunthill in 1992 were considerably higher than in 1993 (Fig. 3-10). There were also different weather patterns in the two years: in 1992, the outbreak site remained clear of snow until mid-December, whilst in 1993 the site was covered by approximately 40 cm of snow from 20 November onwards (Fig. 3-10ii).
Total numbers of trapped adult male O. brumata were marginally higher in 1993 than 1992, although the interpretation of this result is complicated by the fact that the traps were not in place in 1992 until 12 November, by which time a proportion of the male emergence, which occurs earlier than the female emergence (Cuming, 1961), may have been missed.
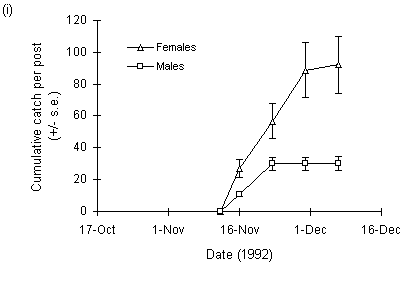
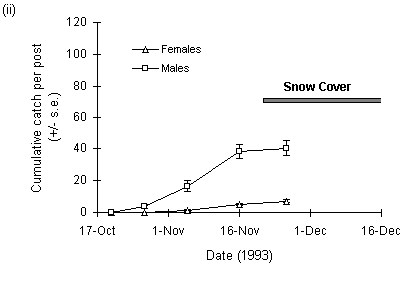
Figure 3-10. Cumulative counts of adult O. brumata caught on
sticky traps at
Hunthill moorland outbreak site in (i) 1992 and (ii) 1993.
Densities of O. brumata larvae in June 1993 and 1994 followed the same pattern as the adult female emergence data from the preceding winter, with considerably higher densities in 1993 than in 1994 (Fig. 3-11i).
At Swartaback, Orkney, larval density was high in both 1994 and 1995 (Fig. 3-11ii), and there was no significant change in density between years (Mann-Whitney U-test, n.s.).
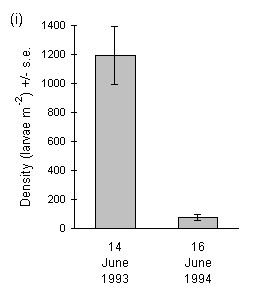
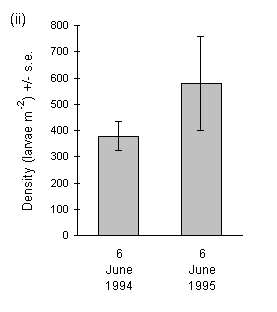
Figure 3-11. Mean larval densities of O. brumata in successive
years at (i) Hunthill (mixed Calluna and Vaccinium vegetation) and (ii)
Swartaback, Orkney (pure Calluna). Note that the Hunthill density value
for 1993 is derived from the data presented previously in Figure 3-5, and
is the mean value for all samples from mixed Calluna-Vaccinium stands
on 14 June.
Developing Vaccinium myrtillus leaf buds were rapidly colonised by newly-emerged O. brumata larvae at Glen Shee in 1993 (Fig. 3-4). This occurred in late April, before Calluna vulgaris at the site had produced any new season's growth. Old Calluna foliage, from previous years' growth, is tougher and less nutritious than new shoots (Moss, 1967). Vaccinium myrtillus at this site, therefore, supported the establishment of early-emerging larvae at a date when no other suitable host was available, a rôle which has previously been proposed by Fielding (1992, Chapter 9).
In contrast to the early pattern of host use, by June 1993 larval densities at Hunthill were generally higher on Calluna than on Vaccinium (Fig. 3-5). By this date, plentiful new growth of Calluna was available, whilst most Vaccinium at the site was heavily defoliated (L.E.B. Kruuk, pers. comm.). Defoliation of Vaccinium during the early O. brumata feeding period may induce larvae to switch from Vaccinium to Calluna, and the observed increase in larval density with time in the pure Calluna vegetation category supports this hypothesis. Mark-recapture studies would be required in order to quantify the extent of larval migration between vegetation classes, although the marking of immature O. brumata larvae might not be feasible. The observed decline in larval densities with time in the other three vegetation classes (Fig. 3-5) can be explained by pupation between sampling dates, predation, and possible emigration of larvae from these areas.
The results of this study clearly confirm the hypothesis that C. vulgaris is a low-quality host for O. brumata. Survival was significantly lower on Calluna than on Vaccinium (Fig. 3-8i), which reflects the difficulty of establishment on a plant which is structurally and biochemically dissimilar to the more usual broadleaved O. brumata hosts. Although the 50% survival rate shown here on Vaccinium might also appear poor, it agrees with the estimate of 50% given by Tammaru et al. (1995) for the similar Geometrid Epirrita autumnata feeding in cages on native birch trees. High larval mortality may be a common feature of E. autumnata and O. brumata, perhaps due to the small size of newly-emerged larvae.
Larval development in the field was significantly retarded in Calluna-dominated vegetation on two sampling occasions (Fig. 3-6). Although the possibility of migration between vegetation classes complicates the interpretation of such field results, larvae constrained by feeding bags also showed significantly retarded development on Calluna compared with that on Vaccinium (Fig. 3-8ii). This agrees with work by Fielding (1992), who observed retarded development of Hydriomena furcata (Lepidoptera: Geometridae) (July Highflyer) on Calluna relative to Vaccinium. Host species had no effect on O. brumata development time over the penultimate and final instars (Section 3.3.4), which suggests that host quality affects larval development most strongly during the early instars. One possibility is that the initiation of first instar feeding is delayed on Calluna due to extended larval searching time. This would result in a developmental lag, but not necessarily a difference in the rate of progress through the subsequent instars, between larvae on different hosts (as found for H. furcata by Fielding (1992)).
The larvae used in the pot feeding trials (Section 3.2.4) were reared on Vaccinium prior to the start of the experiment. Genuine differences in host quality are therefore confounded with possible effects of the dietary change from Vaccinium onto Calluna at the start of the fourth instar. Attempts were made to control for the latter effect by conducting the reciprocal experiment, whereby larvae were initially reared on Calluna. However, it proved impossible to rear stocks of larvae on excised Calluna shoots, due to failure of first instar larvae to commence feeding. This observation reinforces the difference in quality between these two hosts.
A second caveat of the pot feeding trials is the possibility of induced chemical changes in plant tissue resulting from excision (Risch, 1985). However, the greater level of frass production on Calluna than on Vaccinium (Figs 3-9i & ii) suggests that Calluna is less digestible than Vaccinium, and this is consistent with the known high concentrations of indigestible fibre and lignin in Calluna foliage (Iason et al., 1993). Concentrations of these inert compounds are unlikely to have changed significantly in excised host tissue within the time-scale of these feeding trials (Hahlbrock & Scheel, 1989).
Larval growth rates and pupal weights were significantly higher on Vaccinium than on Calluna in the feeding trials (Fig 3-9iii & iv). The same effect on pupal weight was demonstrated in field-sampled populations (Fig. 3-7). However, the pupal weights attained in the feeding trials were substantially greater than those of field-sampled larvae, possibly due to the more protected environment and the regular supply of fresh food experienced by larvae in the feeding trials.
There is normally a close relationship between O. brumata pupal weight, adult female body weight, and potential egg production (Roland & Myers, 1987) (see Fig. 4-8ii). However, this study failed to demonstrate a significant effect of host species on adult female body weight (Table 3-1), despite the clear effect on pupal weight shown above. This could be due to the low statistical power of the analysis used (Section 3.3.2), or alternatively may indicate that potential egg production is not greater after feeding on Vaccinium than on Calluna. In contrast, adult male body weight and body length were significantly greater after feeding on Vaccinium than on Calluna (Table 3-1).
Taken as a whole, these results show that Vaccinium myrtillus is a better host for O. brumata than Calluna vulgaris. The results imply that the presence of Vaccinium enhances O. brumata survival and development on moorlands. The hypothesis that moorland O. brumata populations are constrained by the "bottom-up" effect of host quality is strengthened by the poor larval performance on Calluna and enhanced performance on Vaccinium shown here. The fact that moorland O. brumata outbreaks in mainland Scotland usually occur on Vaccinium-rich sites is consistent with the hypothesis that localised "enrichment" of the moorland vegetation (in this case by the presence of Vaccinium) increases the probability of outbreaks.
However, the near-total absence of Vaccinium from the O. brumata outbreak sites on Orkney (Section 2.3) challenges the above view. Calluna is the only host in most Orkney outbreaks, and the results of this chapter show that O. brumata can complete its life cycle successfully on Calluna alone. Although performance is enhanced on Vaccinium, the assumption that individual insect performance translates directly to population density has already been criticised (see Section 1.2.3). Furthermore, the fact that moorland O. brumata outbreaks appear to be a recent phenomenon (Section 2.1) argues against Vaccinium as a fundamental cause, since it is a native moorland species (Gimingham, 1972). Indeed, its widespread distribution on the moorlands of N.E. Scotland may be a sufficient explanation for the apparent tendency for O. brumata outbreaks there to occur on Vaccinium-rich moorlands. The absence of outbreaks from many other Vaccinium-rich moorlands, and the absence of Vaccinium from the Orkney outbreaks, show that Vaccinium is neither a sufficient cause nor a prerequisite for this phenomenon.
Strikingly different patterns of larval density were observed between the Hunthill and Swartaback moorland outbreaks (Fig. 3-11). At Hunthill, there was a sharp decline in density from an extremely high level in 1993 to approximately 10% of this value in 1994. In contrast, the Swartaback site showed no such decline. The decline at Hunthill was not caused by exhaustion of available host material, since defoliation of Calluna at the site was heavy but not complete at the end of the 1993 feeding season, whilst Vaccinium at the site showed good regrowth in 1994 following its defoliation in 1993 (J.E. Kerslake, pers. obs.). Neither was there any sign of disease in O. brumata larvae at Hunthill in either year.
The decline in larval density at Hunthill followed a similar drop in adult female abundance in 1993, which in turn was associated with deep snow cover of the site (Fig. 3-10ii). Snow disrupts the emergence of O. brumata from its underground pupae (Cuming, 1961), and snow cover at such high-altitude moorland sites is frequently long-lasting (Watson et al., 1994). These data therefore suggest that November snow cover can force moorland O. brumata populations into rapid decline by inhibiting adult emergence. Conversely, O. brumata populations may increase in years of "climatic release" (Section 1.3.2), when early-winter snow cover is absent. If this hypothesis is correct, then low-altitude Orkney O. brumata populations should be less frequently affected, because snow cover is rare on Orkney during November and December (E. Meek, pers. comm.). Consequently, Orkney O. brumata populations should show less sharply fluctuating dynamics than those at high-altitude mainland sites, as found here (Fig. 3-11ii). Other factors, for example parasitism, may be more important than snow cover in limiting O. brumata population levels on Orkney (see Chapters 4 & 8).
The above hypothesis assumes that larval density is limited by the number of eggs laid in the previous winter. There is insufficient evidence at present to determine whether this is the case on moorlands, and longer-term monitoring is clearly necessary to determine whether snow patterns can be used to predict the probability of future O. brumata outbreaks. However, if the model is correct, then the effect of future climatic changes on snow cover (Watson et al., 1994) will have considerable implications for the susceptibility of high-altitude moorlands to such outbreaks.
The occurrence of O. brumata outbreaks on Scottish moorlands is surprising in view of the climatic severity of such sites and the nutritional inadequacy of Calluna vulgaris, the dominant host. This chapter has therefore focused on host quality, host exploitation, and climate.
The results affirm that Vaccinium myrtillus is a better host than C. vulgaris, and that its presence enhances O. brumata survival and development. The presence of Vaccinium may facilitate outbreaks in mainland Scotland.
However, the presence of Vaccinium is not a prerequisite for outbreak occurrence. The Swartaback moorland outbreak, which is typical of those on Orkney, lacks Vaccinium. Low-altitude O. brumata populations on Orkney moorlands may persist, despite the absence of Vaccinium, due to lower probability that adult emergence will be disrupted by snow. Orkney moorland O. brumata populations may exhibit less variable dynamics than their high-altitude mainland counterparts.
† For ease of reading, Vaccinium myrtillus will be referred to in the text as "Vaccinium". Note that the other common moorland Vaccinium species, V. vitis-idaea, is not fed upon by O. brumata, although it is occasionally present in small amounts at outbreak sites.
|
Previous Chapter |
 Contents Page |
Next Chapter |