|
Previous Chapter |
 Contents Page |
Next Chapter |
|
Previous Chapter |
 Contents Page |
Next Chapter |
Descriptions are given of the life history and host range of O. brumata and the ecology of its principal moorland host-plants. The history and characteristics of O. brumata outbreaks on Scottish moorlands are summarised, and the field sites used in this study are described. Previous studies of the effects of insect defoliation on moorland vegetation are reviewed and compared with the effects of O. brumata outbreaks.
Since 1980, Operophtera brumata L. (Lepidoptera: Geometridae), winter moth, has emerged as a serious pest of moorlands in Scotland. Although better-known as a defoliator of broadleaved woodland and orchard trees (Varley & Gradwell, 1968; Tenow, 1972; Holliday, 1977), O. brumata has recently attained outbreak densities on the moorlands of Orkney, (Picozzi, 1981), mainland Scotland (Kerslake et al., 1996), and Shetland (C. Donald, pers. comm.). The principal host-plant used by O. brumata in these moorland outbreaks is Calluna vulgaris (L.) Hull (Ericaceae), common heather, although Vaccinium myrtillus (L.) (Ericaceae), bilberry, is also used if present in the outbreak area. A severe moorland outbreak of O. brumata can result in the defoliation of hundreds of hectares of moorland vegetation (J.E. Kerslake, pers. obs.). This phenomenon is of considerable conservation significance, since Calluna-dominated moorland is internationally rare (Ratcliffe & Thompson, 1988), and its regeneration following an O. brumata outbreak is frequently poor (Section 2.3.1).
The occurrence of moorland O. brumata outbreaks represents the second major change in the feeding patterns of this insect in Scotland in recent years, since O. brumata has also recently expanded its host range to include Picea sitchensis Bong., Sitka spruce, a non-native conifer planted in abundance in Scotland since the 1950s (Stoakley, 1985; Fraser, 1995). In the present case, although Calluna vulgaris is a native species on which O. brumata has been known to feed for many years (Traill, 1869), severe outbreaks of O. brumata on moorlands were unrecorded in the scientific literature prior to 1981 (Picozzi, 1981). The recent, apparently unprecedented examples of moorland O. brumata outbreaks in Scotland are surprising because C. vulgaris is likely to be a poor-quality host (see Section 2.2.2 ).
In this chapter, the host range and life history of O. brumata are described, followed by the ecology of C. vulgaris and other associated moorland plant species. Descriptions are given of the history and characteristics of O. brumata outbreaks on moorland vegetation in Scotland, and of the field sites at which moorland and woodland outbreak populations of O. brumata were studied. The mechanisms by which insect outbreaks can cause the loss of moorland vegetation are then discussed in more detail, with reference to recent studies of Lochmaea suturalis (Thompson) (Coleoptera: Chrysomelidae), heather beetle, another eruptive moorland herbivore.
Operophtera brumataå is a polyphagous univoltine moth, common in Britain and throughout much of Europe, from northern Scandinavia to southern Italy (Wylie, 1960a; Holliday, 1985). It is an economically important pest of a range of broadleaved tree species, including oak (Quercus spp.) (Gradwell, 1974), birch (Betula spp.) (Tenow, 1972), willow (Salix spp.) (Kirsten & Topp, 1991), hazel (Corylus avellana L.) (Wint, 1983a) and apple (Malus spp.) (MacPhee et al., 1988), and is also common on a range of moorland shrubs, including bilberry (Vaccinium myrtillus) (Tenow, 1972; Fielding, 1992), common heather (Calluna vulgaris) (Traill, 1869) and bog myrtle (Myrica gale L.) (Stoakley, 1985). Outbreaks of O. brumata are common on many of the above tree species: Operophtera brumata, together with the ecologically similar Geometrid species Operophtera fagata and Oporinia (=Epirrita) autumnata, frequently defoliates large areas of birch forest in the Scandinavian mountain chain, and these synchronised multi-species outbreaks have an approximate ten-year periodicity (Tenow, 1972; Niemelä, 1980). During the 1950s, accidental introduction of O. brumata to Nova Scotia resulted in outbreaks and widespread defoliation of urban shade trees, red oak (Quercus rubra) and apple orchards (Embree, 1965; MacPhee et al., 1988), and a similar phenomenon occurred following the introduction of O. brumata to British Columbia circa 1970 (Roland & Embree, 1995). Outbreaks of O. brumata have also recently damaged Sitka spruce (Picea sitchensis) plantations in Scotland (Fraser, 1995).
Operophtera brumata overwinters as an egg. In woodland systems, eggs are laid in crevices in the twigs, branches or main stem of the host tree during November and December (Cuming, 1961; Watt et al., 1992), and are extremely cold-resistant (MacPhee, 1967). It is unlikely that O. brumata oviposits selectively on nutritionally favourable host-plants (Price et al., 1990; Renwick & Chew, 1994), since adult O. brumata do not feed, the females are flightless (Cuming, 1961), and many hosts are leafless at the time of oviposition. However, Graf et al. (1995) found a slight preference for females to climb cherry (Prunus spp.) over apple.
The eggs of O. brumata are ovoid, approximately 0.6 mm in length, and are initially green, but fertilised eggs change to an orange-brown colour several days after they are laid. Mature eggs assume a blue-black colour approximately two days before hatching. Hatching date varies from early March (in southern Italy) to May (in Norway) (Holliday, 1985), but commonly occurs in synchrony with the budburst of the predominant host-plant (Feeny, 1970; Holliday, 1977; Hunter, 1992) (see Section 1.2.4). Speyer (1941) and Wylie (1960a) showed by means of cross-breeding experiments that the duration of the egg stage is partly genetically determined, although egg development time is also inversely related to temperature (Wylie, 1960a; Embree, 1970). On broadleaved hosts, newly-emerged larvae burrow into the developing leaf buds, where they feed inside shelters constructed by spinning together the developing leaves with silk. Newly-emerged larvae are approximately 1 mm in length, can survive for around five days without food, and those which are initially unsuccessful in locating suitable leaf buds disperse on the wind by means of long silk threads (Cuming, 1961; Edlant, 1971).
Feeding larvae pass through five instars, characterised by five distinct head capsule widths (Cuming, 1961), after which they cease feeding for several days, empty their guts and drop on silk threads to the ground. They then burrow beneath the surface to a depth of between 5 and 12 cm and pupate in silken cocoons, a process which takes approximately six days (Cuming, 1961). Formation of the pupa is normally completed by early July, although parasitised larvae may develop more slowly than unparasitised ones (J.E. Kerslake, pers. obs.) (see Section 1.3.2 for further discussion of the effects of parasitoids on O. brumata).
The date of adult emergence varies from late September (in Russia) to February (in Sicily) (Uvarov, 1931; Holliday, 1985), and variation between populations in the duration of the pupal stage is heritable (Speyer, 1941). Schneider-Orelli (1916) showed that the date of adult emergence is earlier at high altitudes, possibly as a result of selection for avoidance of winter weather conditions, since snow can disrupt adult emergence (see Chapter 3). Peak abundance of adult female O. brumata was recorded on 27 November in Kent by Briggs (1957) (but see Chapter 4 for emergence dates in Scottish moorland and woodland populations). Males emerge approximately three days before females (Briggs, 1957; Cuming, 1961), a strategy known as "protandry", which is believed to maximise reproductive success in both males and females (Fagerström & Wiklund, 1982). Operophtera brumata is brachypterous, and the flightless adult females climb tree trunks or other nearby tall objects at dusk, and release pheromone. Shortly afterwards, males fly and locate the females, and mating takes place. Both males and females may mate more than once (Cuming, 1961). Oviposition by caged O. brumata usually occurs within 48 hours of mating (J.E. Kerslake, pers. obs.)
Calluna vulgaris, common heather or ling, is a woody ericoid evergreen dwarf shrub (Gimingham, 1960), which forms the dominant vegetation over extensive areas of upland heathland in Europe (Gimingham, 1972). On exposed, upland moorlands it commonly adopts a dwarfed, prostrate or "procumbent" growth habit, which is partially heritable (Gimingham, 1960). Calluna vulgaris has been present in Britain from late glacial times (Gimingham, 1960), although Calluna-dominated heathland is currently an internationally rare and diminishing habitat type (Marrs, 1986; Marrs et al., 1986; Ratcliffe & Thompson, 1988; Berdowski, 1993).
The growth of new season's C. vulgaris shoots commonly begins in April or May (Gimingham, 1960), and current season's growth is more nutritious for birds than the previous year's foliage (Moss, 1967). Nevertheless, C. vulgaris foliage contains high levels of tannins, lignin and fibre (Iason et al., 1993) and relatively low concentrations of tissue nitrogen (Moss, 1967; Iason et al., 1993; Pitcairn et al., 1995), compared with the young foliage of many broadleaved tree species (Feeny, 1970; Wint, 1979) and is consequently likely to be a poor-quality food source for a generalist Lepidopteran such as O. brumata.
Although heathland vegetation is frequently dominated by C. vulgaris, other associated plant species are also common. For example, Vaccinium myrtillus (bilberry), a deciduous dwarf shrub, is often found in association with C. vulgaris in upland heaths on well-drained acid soils (Ritchie, 1956; Gimingham, 1972). Vaccinium myrtillus initiates new growth earlier in the season than C. vulgaris, in late March to April (Gimingham, 1972; Lähdesmäki et al., 1990), and has higher foliar nitrogen concentrations than C. vulgaris during early summer (Powell & Malcolm, 1974; Lähdesmäki et al., 1990). Other plant species which commonly occur in association with C. vulgaris on moorlands in Scotland include Erica cinerea (bell heather), Empetrum nigrum (crowberry), Vaccinium vitis-idaea (cowberry) and Nardus stricta (mat grass) (Gimingham, 1972).
The following description of moorland O. brumata outbreaks is derived from personal observations, supplemented with information provided by officials of Scottish Natural Heritage (Liz McTeague, Peter Reynolds and Angus MacDonald) and the Royal Society for the Protection of Birds (Eric Meek). Further information was gained from conversations with local land-owners and gamekeepers.
The first record of an outbreak of O. brumata on C. vulgaris was made by Picozzi (1981), who noted a mixture of both O. brumata and Agriopis marginaria (Lepidoptera: Geometridae) (dotted border) larvae, at a total density in excess of 1500 larvae m-2 on an area of moorland at Queenamidda on mainland Orkney (O.S. HY 371210) in June 1980. Since this date, outbreaks of O. brumata (with or without associated species) have occurred on at least three other moorland sites on mainland Orkney ć, on Shetland (C. Donald, pers. comm.) and on at least four moorland sites on mainland Scotland (J.E. Kerslake, pers. obs.). Although O. brumata has been the most destructive outbreaking Lepidopteran species on Scottish moorlands since 1980, outbreaks of several other species have also caused similar defoliation to C. vulgaris: these species include Agriopis aurantiaria (Geometridae) (scarce umber) (S.E. Hartley, pers. comm.), Agriopis marginaria (Picozzi, 1981), Orgyia antiqua (Lymantriidae) (vapourer moth) and Abraxas grossulariata (Geometridae) (magpie moth) (A. MacDonald, pers. comm.). In addition to the above species, Hydriomena furcata (Geometridae) (July high flyer) and Epirrita spp. are also commonly observed in association with O. brumata in its moorland outbreak sites, but always at lower densities than O. brumata (J.E. Kerslake, pers. obs.).
The majority of moorland O. brumata outbreaks in mainland Scotland have occurred at altitudes of 500 to 650 m above sea level (Plates I & II), typically on exposed slopes with mature stands of Calluna growing on freely-drained shallow peat (although the Calluna is often dwarfed by the harsh climates at these altitudes (Gimingham, 1960)). Such sites are seldom burnt by moorland managers, due to their relative inaccessibility. Estate workers have noted a tendency for outbreaks to be most severe on those sides of the hills which face into the prevailing winds, with the more sheltered hillsides less affected, although this observation has not been rigorously tested. Estate workers also consider that outbreaks are more severe in years of warm, dry weather during May and June, although again this is an untested observation. Vegetation at these outbreak sites is usually dominated by bushy patches of Calluna vulgaris and of Vaccinium myrtillus, with occasional clumps of Erica spp., Empetrum nigrum, Vaccinium vitis-idaea and Nardus stricta. However, O. brumata only defoliates Calluna and V. myrtillus at such sites, and other plant species are left almost totally untouched, even when surrounded by completely defoliated Calluna.
The O. brumata outbreak sites on Orkney contrast greatly with the above description of mainland sites. On Orkney, the affected moorlands are typically low-altitude (20 to 50 m above sea level), flat or gently sloping stands of dominant Calluna, with negligible amounts of V. myrtillus or of the other moorland species mentioned above (Plate III). Calluna in these sites is lush, mature and up to 1 metre tall (R.S.P.B. has a policy not to burn such sites, in order to maintain cover for nesting birds).
Larval densities can be extremely high in moorland outbreaks, and O. brumata larvae are clearly visible feeding on Calluna shoot tips (Plate IV), which they spin together with silk, during late May and June. The high density of larvae attracts avian predators, and outbreaks are frequently accompanied by flocks of feeding gulls and crows (Picozzi, 1981). At high densities, approximately 50% of the feeding larvae resemble the "black-green" coloration described by Tenow (1972), although O. brumata larval colour can change between instars, and intermediate colours between the extreme "dark" and "light" forms observed by Tenow (1972) are common in moorland outbreaks (J.E. Kerslake, pers. obs.).
Defoliation of C. vulgaris (and, if present, V. myrtillus) by a severe O. brumata outbreak can be almost total. Both the current season's Calluna foliage and the older growth from previous years is eaten, although the more nutritious current season's growth (Moss, 1967) is consumed first. Estate workers have noted that the top shoots of defoliated Calluna plants begin to die back during late June, and by August the stems of affected Calluna plants assume a characteristic "fox-red" colour. By the following spring, dead grey Calluna shoots dominate the affected area (a more detailed description of the mechanisms which lead to the death of Calluna is given in Section 2.3.3). Vaccinium myrtillus is more resistant to defoliation, and often shows good regrowth in the year following an outbreak.
Regeneration of Calluna is poor after an O. brumata outbreak in mainland sites, possibly due to heavy grazing pressures in these areas. The vegetation at such sites is rapidly dominated by Nardus stricta and Deschampsia flexuosa. Total losses of Calluna-dominated heathland vegetation in two severely-affected mainland sites have exceeded 400 ha at each site (A. MacDonald, pers. comm.; J.E. Kerslake, pers. obs.). In contrast, some regeneration of Calluna has occurred at the Queenamidda site on Orkney (Hartley & Kerslake, 1994), by vegetative regrowth from the bases of the old stems, and this regrowth should eventually be sufficient to restore the Calluna canopy. Regenerating Calluna at Queenamidda benefits from low grazing pressure and lack of competition from pre-established grass species (Hartley & Kerslake, 1994).
Insufficient data currently exist to determine whether O. brumata outbreaks commonly persist at a site for many years, or whether they are short-lived phenomena. Anecdotal evidence from estate workers, combined with personal observations, suggests that high larval densities often persist for two or three years, but severe outbreaks are unlikely to persist for more than three years in a single location. Chapter 3 presents data and further discussion of O. brumata population levels in moorland outbreaks.
Adult emergence of O. brumata on moorlands in mainland Scotland occurs mostly during November (see Section 4.3.2 for data). Male moths climb to the top shoots of Calluna plants shortly before dusk, particularly after a warm day, and hundreds of them may be disturbed by walking through the outbreak area at this time. The flightless females also climb to the top shoots at dusk, although it is not known whether they release pheromone in the same way as in woodland populations (Section 2.2.1), or whether the pheromone is effective on exposed moorlands. On still evenings, a mass flight of males takes place, starting at dusk and lasting approximately five minutes. Males fly no more than 1 metre above the top of the Calluna canopy, then re-alight onto the vegetation. After this event, pairs of mating moths can readily be found on the Calluna stems. The favoured oviposition site for O. brumata in moorland vegetation is not known, although eggs have occasionally been observed in crevices of the woody Calluna stems. Hatching dates of moorland O. brumata populations are investigated in Chapter 5.
Figures 2-1 and 2-2 show the locations of the principal O. brumata outbreak sites used in this study. Descriptions of these sites are given below:
Swartaback moorland, Orkney (Fig. 2-1i), HY 376077, alt. 30 m.
This outbreak site on mainland Orkney occupied approximately 5 hectares of flat Calluna-dominated moorland adjacent to the Loch of Kirbister (Fig. 2-1i). None of the other moorland plant species listed above (Sections 2.2.2 & 2.3.1) were noted at the site, and the Calluna canopy was thick, mature and tall (approximately 80 cm), providing shelter for large numbers of breeding birds (E. Meek, pers. comm.). The vegetation at this site was not regularly burned in the years prior to the outbreak, but is currently subject to controlled grazing by sheep.
Hunthill moorland, N.E. Scotland (Fig. 2-1ii), NO 464755, alt. 640 m. (Plate I)
This O. brumata outbreak site occupied an exposed, south-east facing ridge of mixed C. vulgaris-V. myrtillus moorland, near to the summit of Black Hill (Fig. 2-1ii).
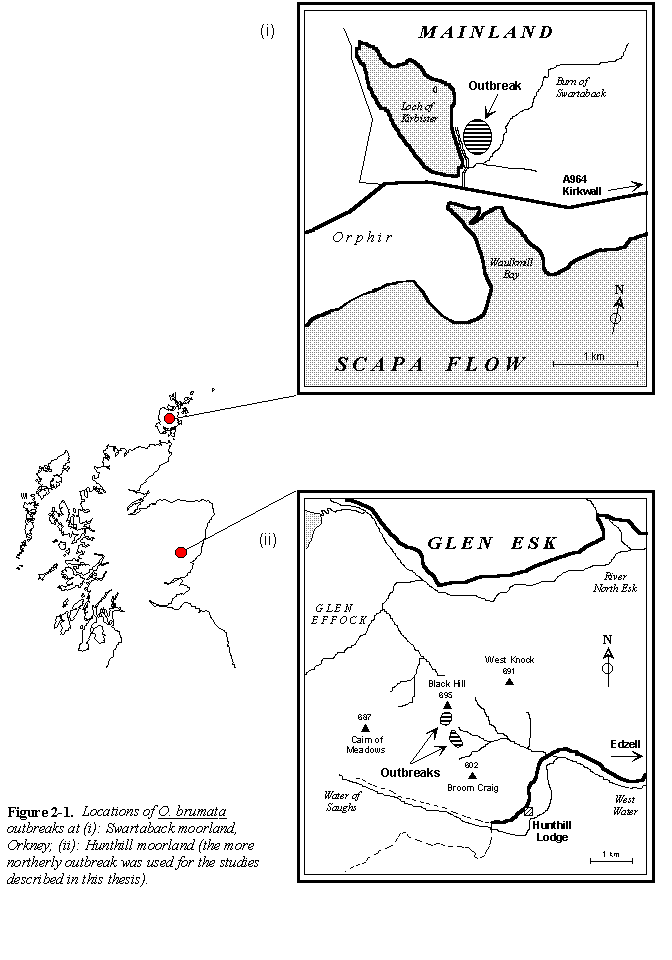
The outbreak area was small (approximately 2 hectares), but a second area of O. brumata infestation was also noted on an east-facing valley side just below the Black Hill site (Fig. 2-1ii), and several other outbreaks on the surrounding hills were also reported by staff of the Hunthill estate. An outbreak was also reported to have occurred on a considerably lower-altitude area of moorland on the Hunthill estate (NO 462711 alt. 330 m.) but visits to this site in winter 1992 and spring 1993 failed to find any signs of O. brumata or other outbreaking insects. Estate staff reported no evidence that plantations of Picea sitchensis at altitudes of approximately 300 m on Hunthill estate had ever suffered outbreaks of O. brumata.
Bonar Bridge oak woodland (Fig. 2-2i), NH 660889, alt. 50 m.
This area of primarily Quercus petraea woodland adjacent to the Dornoch Firth was severely defoliated by a mixed-species insect outbreak (with O. brumata the most abundant of these species) in 1993 and again in 1994. A third visit to the site in June 1995 found low levels of defoliation, and no feeding O. brumata larvae.
The total outbreak area in 1993 and 1994 was not determined, but included extensive stands of Q. petraea on both sides of the A9 road (Fig. 2-2i).
Glen Shee moorland, N.E. Scotland (Fig. 2-2ii), NO 142765, alt. 630 m. (Plate II)
The area used for the present study (Chapter 3) was a small (approx. 2 hectares) outbreak of O. brumata (with Hydriomena furcata also abundant) on mixed C. vulgaris-V. myrtillus vegetation, with other associated moorland plant species (Section 2.3.1). The outbreak area occupied an east-facing slope adjacent to the A93 road (Fig. 2-2ii), and densities of O. brumata were high at the site in winter 1992 and spring 1993 (Chapter 3), but declined thereafter. Considerably larger areas of moorland to the east of this site were also infested with O. brumata between 1990 and 1993 (see Fig. 2-2ii). Although the total area affected by O. brumata outbreaks at Glen Shee was not determined during the course of this study, gamekeepers estimate it to be several hundred hectares. An outbreak of Agriopis aurantiaria also occurred on moorland at Glen Shee in 1993 (S.E. Hartley, pers. comm.).
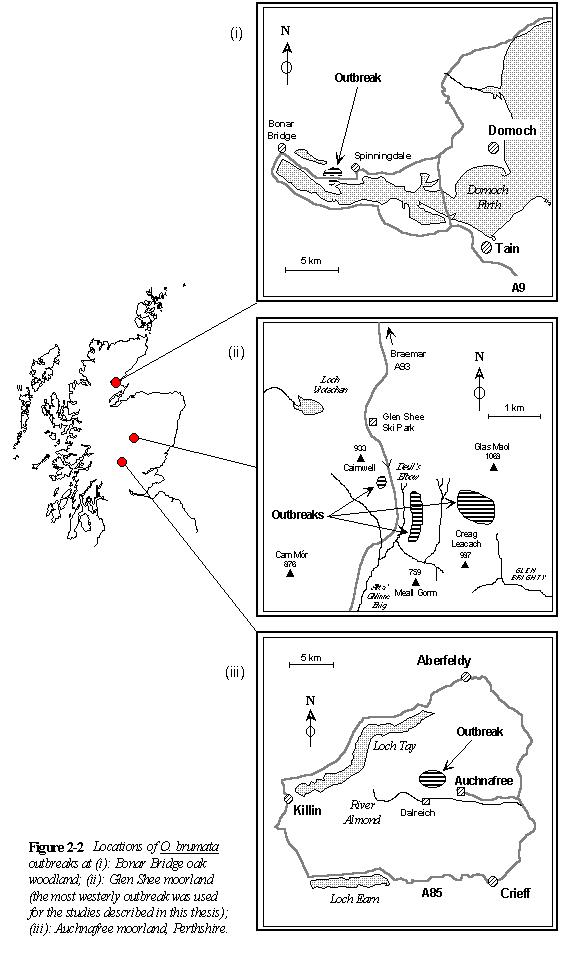
Auchnafree moorland, Perthshire (Fig. 2.2iii), NN 7734, alt. 450-650 m.
Operophtera brumata outbreaks have occurred in several locations on the Auchnafree estate, and losses of Calluna-dominated vegetation are currently estimated by the gamekeeper to exceed 400 ha. The area indicated in Figure 2-2iii is the most heavily-affected part of the estate. Estate workers have noted that the losses of Calluna have been most severe on west-facing hillsides, and the prevailing winds on this estate come from the west. The most severe outbreaks have occurred on exposed ridges, near the tops of these hills, at altitudes between 450 and 650 m above sea level. Die-back of Calluna at Auchnafree was first noted by the gamekeeper around 1981, but was initially mistakenly attributed to the effects of "acid rain"; Operophtera brumata was identified as the actual cause in 1991. Vegetation at the O. brumata outbreak sites is a mosaic of C. vulgaris and V. myrtillus patches, with occasional associated species as described above (Section 2.3.1).
In order to understand in more detail the mechanisms by which insect outbreaks can result in the loss of moorland vegetation, it is necessary to review recent studies of another eruptive moorland pest, Lochmaea suturalis, the heather beetle, outbreaks of which are common on heathlands in Europe. A recent increase in the frequency and severity of L. suturalis outbreaks in Holland has been attributed to nitrogen enrichment of heathland vegetation there, caused by air pollution (Heil & Diemont, 1983; Berdowski, 1993) (see Section 1.2.3). Some key characteristics of L. suturalis outbreaks and their effects on heathland vegetation are presented here, and their similarities or differences with O. brumata outbreaks are highlighted. Any unattributed information in this section was obtained from the thorough review of Dutch heathland dynamics by Berdowski (1993).
Lochmaea suturalis, unlike Operophtera brumata, is monophagous on Calluna vulgaris. Eggs are laid between April and June, often in moist Sphagnum moss at the base of the Calluna stems (Cameron et al., 1944; Scandrett & Gimingham, 1991). Larvae of L. suturalis emerge in June (later than those of O. brumata; Section 2.2.1) and feed on young Calluna shoots, passing through three instars and finally pupating beneath the soil or moss in late July or early August (for a review of the whole life cycle, see Cameron et al., 1944). Defoliation of C. vulgaris by L. suturalis is most severe during the larval feeding period, and causes the Calluna stems and remaining leaves to assume a striking "fox-red" colour in July and August (similar to that seen in moorland O. brumata outbreaks; Section 2.3.1), and which is visually suggestive of severe drought stress. However, there is no sustained reduction in C. vulgaris xylem water potential at this stage, which implies that plants are not severely water-stressed in the short term (Berdowski, 1993).
The longer-term effects of defoliation by L. suturalis on C. vulgaris physiology are, however, more damaging. Penetration of air from wounded leaves into the xylem vessels eventually blocks water transport and causes tissue death several months after defoliation (Berdowski, 1993). In addition, loss of photosynthetic production due to defoliation prevents the usual accumulation of carbohydrate reserves in the fine roots during the growing season; this in turn inhibits root respiration, causes a breakdown in root structures over the winter, and prevents adequate root activity to sustain shoot regrowth in the next season (Berdowski, 1993). Death of above-ground C. vulgaris tissues therefore occurs over winter or in early spring, due to defoliation in the previous summer. This pattern of C. vulgaris death is extremely similar to that observed following moorland O. brumata outbreaks in Scotland (Section 2.3.1), although to date no studies have tested whether the same causal mechanisms apply.
Dead grey Calluna shoots are often conspicuous in areas affected by L. suturalis in the year following defoliation (Cameron et al., 1944), and the resultant opening of the Calluna canopy allows greater penetration of light to ground-level vegetation. This benefits grasses such as Deschampsia flexuosa and Molinia caerulea, which can survive beneath a closed Calluna canopy, but which require a gap in this canopy before they can become dominant (Berdowski, 1993). Lochmaea suturalis outbreaks therefore favour a change in vegetation type from Calluna-dominated heathland to grassland (Heil & Diemont, 1983; Heil & Bobbink, 1993). This is again extremely similar to the effect of a moorland O. brumata outbreak (Section 2.3.1), although in north-east Scotland Nardus stricta is the most usual grass species to benefit from the open Calluna canopy (J.E. Kerslake, pers. obs.).
Regeneration of C. vulgaris can occur from seed, if competition from other vegetation types does not prevent establishment, or by vegetative regrowth from the basal stems of older plants. However, the regenerative abilities of Calluna stems decline with age, and are further reduced by nutrient enrichment (Berdowski & Siepel, 1988).
Lochmaea suturalis has been known as a pest of moorlands in Britain for many years (Grimshaw, 1898). Outbreaks of L. suturalis, apparently triggered by a succession of years with high spring precipitation, caused extensive damage to Scottish moorlands between 1933 and 1937 (Cameron et al., 1944). More recently, Marrs (1986) observed a transition from Calluna to Betula-documented vegetation in an East Anglian lowland heath following a combination of drought and L. suturalis outbreak. However, there is currently no published evidence for a recent increase in the frequency or severity of L. suturalis outbreaks in Britain, such as that seen with O. brumata in Scotland or with L. suturalis in Holland.
In summary, L. suturalis outbreaks show many similarities to O. brumata moorland outbreaks (Section 2.3.1). Despite the differing life histories and host ranges of the insects, their physical effects on C. vulgaris and the resultant changes in moorland vegetation are extremely similar. The Dutch work described above provides an insight into the mechanisms by which insect outbreaks affect moorland vegetation dynamics, and is therefore of relevance to the present study. The prospect that Scotland might experience losses of heathland due to O. brumata outbreaks on a similar scale to those caused by L. suturalis in Holland, where 80 km2 of heathland has been lost to grasses in the past four decades (Heil & Diemont, 1983), is clearly an important conservation issue (Ratcliffe & Thompson, 1988). The hypothesis that moorland O. brumata outbreaks in Scotland are caused by a similar mechanism of host-plant nitrogen enrichment to that proposed for L. suturalis in Holland (Heil & Diemont, 1983) was introduced in Section 1.2.3, and is investigated in Chapter 7.
å still referred to in many east-European papers as Cheimatobia brumata
ć further details of affected sites on Orkney may be obtained from the S.N.H. area office, 54/56 Junction Road, Kirkwall.
|
Previous Chapter |
 Contents Page |
Next Chapter |