 Contents Page |
Next Chapter |
 Contents Page |
Next Chapter |
Potential causes of insect outbreaks are discussed; "bottom-up" effects of host-plant quality are contrasted with "climatic release" mechanisms and "top-down" effects of natural enemies. The concepts of nutritional compensation and host race formation are introduced, and the effects of carbon dioxide enrichment on insect-plant interactions are reviewed.
This thesis is concerned with recent outbreaks of Operophtera brumata L. (Lepidoptera: Geometridae), winter moth, on Scottish moorlands. In this chapter, a general introduction is provided to the concepts and hypotheses which are central to the later studies. A detailed description of moorland O. brumata outbreaks is then given in Chapter 2.
Attention is focused initially upon a debate that is central to the current understanding of insect outbreaks in general, and the present phenomenon in particular, namely the importance of host-plant nutritive quality in determining insect abundance. Section 1.2 reviews the argument that phytophagous insects are greatly affected by the nutritional quality of the plants which they eat, and that outbreaks are therefore caused exclusively by increases in this quality. Section 1.3 presents an opposing argument, that outbreaks are not always caused by increased plant quality, but can be caused by "releases" from other constraints, such as climate, or the "top-down" effects of predators and parasitoids. Sections 1.4 to 1.6 then provide introductions to the concepts of nutritional compensation, host race formation and the potential effects of atmospheric carbon dioxide enrichment on insect-plant interactions. Finally, Section 1.7 gives a brief outline of the studies presented in later chapters.
Southwood (1973) pointed out that herbivory is a difficult and relatively specialised evolutionary strategy for insects, since it demands that they convert plant tissue (containing approximately 25% protein by dry weight) to insect tissue (approximately 50% protein). As a consequence, nitrogen for protein synthesis is considered to be a limiting element in the diet of many phytophagous insects (Slansky & Feeny, 1977; White, 1993). The natural environment is variable with respect to the spatial and temporal distribution of available nitrogen (Feeny, 1970; Wint, 1983b; White, 1993), and the successful development and reproduction of an insect herbivore therefore depends on its ability to locate nitrogen in space and time (e.g. Hartley & Gardner, 1995). Outbreaks can be seen as unusually dramatic successes in this endeavour, and so may be made more likely by any factors which increase the availability of nitrogen to insect populations. Such factors include increased leaf nitrogen concentration, or decreased levels of plant secondary compounds which restrict the ability of herbivores to utilise foliar nitrogen (Feeny, 1968, 1976; Wint, 1983b). Mechanisms which can bring about such improvements in the nutritional quality of plant tissue include environmental stresses, and nitrogen deposition from air pollution. In addition, life history adaptations in phytophagous insects can improve their ability to locate high-quality food in space and time. The following sections outline the argument that these "bottom-up" effects of diet quality play an important rôle in the development of insect outbreaks.
One of the earliest and most frequently-cited theories linking insect outbreaks to environmentally-induced changes in host quality is White's "plant stress hypothesis" (White, 1969, 1974, 1976, 1978, 1984). This hypothesis stemmed originally from work on psyllids feeding on Eucalyptus in Australia (White, 1969), but was subsequently extended to include outbreaks of Lepidoptera (White, 1974). The hypothesis states that environmental stresses, such as drought, soil waterlogging or soil nutrient deficiency, can cause biochemical changes in leaf tissue, most importantly an increase in soluble nitrogen concentration; increased leaf nitrogen content then improves insect survival and development on these plants, thereby increasing the probability of an outbreak. White (1984) gave an extensive list of such potential "stresses" which can have the above effect on plant biochemistry, and recently generalised his hypothesis into the extremely broad assertion that "all kinds of outbreaks... have the same basic cause. All are the result of the inevitable evolved response of replicating life forms to increased access to nitrogenous food" (White, 1993).
White's theory has received some anecdotal and experimental support. Niemelä (1980) found that outbreaks of Epirrita autumnata Borkhausen (Lepidoptera: Geometridae) and associated Geometrid species in the Fennoscandian birch forests (see Section 2.2.1) were correlated (although statistically not significantly) with periods of low summer temperatures. On the basis of this observation, Niemelä (1980) argued that cool summers constitute a "stress" (sensu White, 1974), and cause a decrease in the synthesis of phenolic secondary compounds by birch (Haukioja, 1980), thereby increasing foliage quality for insect herbivores. Thomas & Hodkinson (1991) showed that experimental drought stress of Betula pendula and Salix cinerea produced increases in leaf soluble nitrogen concentration, as predicted by White (1974), and provided some evidence that the digestive efficiency of three Lepidopteran species and the biomass gain of one of these three species (Phalera bucephala L. (Lepidoptera: Notodontidae)) were increased in feeding trials on droughted foliage.† Louda & Collinge (1992) showed that severing the roots of Cardamine cordifolia (bittercress) resulted in increased concentrations of both nitrate-nitrogen and the amino acid isoleucine (but not total free amino nitrogen) in the leaves, and increased herbivory by chewing and leaf-mining insects. Myers & Post (1981), in a study of Tyria jacobaeae L. (cinnabar moth) populations on Senecio jacobaea (tansy ragwort), demonstrated that host nitrogen content correlated positively with both the coefficient of variation of moth population density and with larval survival, suggesting that nitrogen-rich plants are particularly prone to outbreaks, as predicted by White (1993).
Other studies, however, have challenged the plant stress hypothesis. Panolis flammea (D&S) (Lepidoptera: Noctuidae) (pine beauty moth) outbreaks in Scotland are associated with Pinus contorta (lodgepole pine) growing in deep unflushed peat, where the poor growth of these trees suggests that they are stressed (Leather & Barbour, 1987). However, Watt (1988, 1989) showed that the survival and growth of caged P. flammea larvae was no better, and was sometimes worse, on the foliage of "susceptible" P. contorta trees than on those from a "resistant" better-drained site, or on Pinus sylvestris (L.) (Scots pine), a host species on which P. flammea outbreaks have never been recorded in Scotland.
Larsson (1989) reviewed the results of experimental tests of White's "plant stress" hypothesis, and concluded that there was "surprisingly little support" for it, particularly among leaf-chewing insects. Waring & Cobb (1992) reviewed 174 studies and concluded that whilst experimental water stress produces negative effects on insect herbivory in a majority of cases, natural droughts generally have the opposite effect. Waring & Cobb (1992) argued that there is a need for a better understanding of the mechanisms of plant responses to environmental stresses, whilst Larsson (1989) drew attention to the lack of consensus on a workable definition of "stress" in this context, and Crawley (1986) considered that plant stress (sensu Grime, 1979) was "not measurable". The problem of definition is reinforced by Price's proposal of an opposing "plant vigour hypothesis" (Price, 1991), based on the observation that many insect herbivores preferentially attack young and "vigorous" plants (e.g. Karban, 1990). Since definitions and measurements of both "stress" and "vigour" are frequently species-specific (e.g. the use of Salix lasiolepis leaf abscission by Preszler & Price (1995)) it is important to focus attention on the more fundamental question, namely whether enrichment of host-plant nitrogen content (whether it be related to "stress", "vigour", or any other such perceived state) causes insect outbreaks.
Many studies have investigated the responses of herbivorous insects to nitrogen enrichment of their hosts. The majority of these studies have shown positive effects (e.g. Taylor, 1988; Port et al., 1995), although this generalisation masks considerable diversity in both insect and plant responses (Mattson, 1980; Waring & Cobb, 1992). Amongst chewing insects, whilst approximately 60% of the studies reviewed by Waring & Cobb (1992) showed a positive response to nitrogen fertilisation, a further 20% showed no response (Fig. 1-1). Insect feeding strategy can affect the response to fertilisation; for example, Feller (1995) found that two specialist herbivores were more responsive than a guild of generalist chewing insects to fertilisation of Rhizophora mangle (red mangrove), and Cockfield (1988) argued that the responses of highly specialised insects to relatively crude manipulations of overall nitrogen availability are harder to predict than those of generalists.
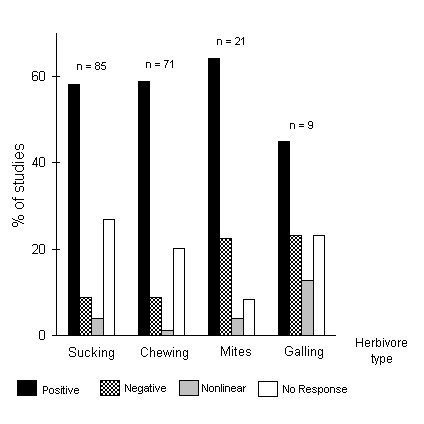
Figure 1-1. Responses of four types of insect herbivore to nitrogen fertilisation of their host-plants, expressed as percentage of studies reviewed. Data and figure redrawn from Waring & Cobb (1992).
Herbivore responses also vary on different host types; Waring & Cobb (1992) found no evidence for a consistent positive response of conifer-feeding insects to nitrogen fertilisation of their hosts, in contrast to the predominantly positive results obtained with insects feeding on broadleaved trees and herbs (Fig. 1-2).
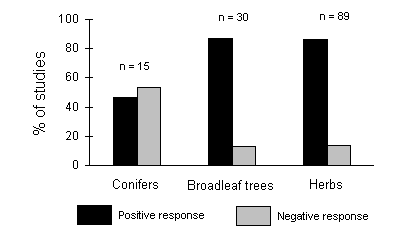
Figure 1-2. Percentage of studies in which insect herbivores responded positively or negatively to nitrogen fertilisation in conifers, broadleaf trees and herbaceous plants. Data and figure redrawn from Waring & Cobb (1992).
The above result (Fig. 1-2) is consistent with the work of Hunter et al. (1991) who studied the development of Operophtera brumata larvae on potted Picea sitchensis (Sitka spruce) trees, and found no significant enhancement of larval survival or growth in response to fertilisation.
In considering the above results, it is necessary to bear in mind the nature of the herbivore "response" in question. The review of Waring & Cobb (1992) pooled the results from studies of growth rate and survivorship at the individual insect level with those of population density responses, the inherent assumption being that individual insect performance translates directly to population density. This assumption is seldom explicitly stated (for discussion, see Fowler & Lawton, 1985; Larsson, 1989), but work by Watt (1988, 1989) on Panolis flammea in Scotland (discussed above, Section 1.2.2) is noteworthy for demonstrating that insect performance sometimes bears little relation to ultimate population levels; in feeding trials, the performance of P. flammea was worst on the plants which are most susceptible to its outbreaks.
Some studies have assessed insect herbivore responses to host-plant fertilisation at the population level. Port et al. (1995) showed that the number of leaf mines of Rhynchaenus fagi L. (beech weevil) on Fagus sylvatica (beech), and the population density of Euceraphis betulae Koch (a birch-feeding aphid) on Betula spp. (birch), were both increased by nitrogen fertilisation of open-air potted trees which were naturally colonised from the surrounding vegetation. Hartley & Gardner (1995) showed a positive response of Philaenus spumarius (Homoptera: Cercopidae) population density on moorland plots of Calluna vulgaris to the application of a nitrogenous fertiliser. However, in the latter study, the observed increase was attributed more to insect immigration and host-plant selection than to increased survival or reproduction in situ on the nitrogen-enriched plants.
The responses of natural systems to atmospheric pollution often provide large-scale "natural experiments" which can help to assess the effects of nutrient enrichment at the population level. For example, Port & Thompson (1980) studied insect abundance on trees and shrubs planted along motorways in the U.K., and recorded outbreaks of Phalera bucephala (Lepidoptera: Noctuidae) (buff tip moth) on Fagus sylvatica and high densities of Euproctis similis (Lepidoptera: Lymantriidae) (gold tail moth) on Crataegus monogyna in these roadside locations. Port & Thompson (1980) also showed that the leaf nitrogen content of roadside plants was high, probably due to emission of nitrogen oxides from car exhausts, and attributed the observed increases in insect abundance primarily to this enrichment (although there was evidence in the same study that insects in isolated traffic islands also benefit from reduced exposure to natural enemies). Witkowski et al. (1992) found an increase in the abundance of leaf miners and galls on Quercus robur L. trees with decreasing distance from a Polish ironworks, together with an increase in the nitrogen-to-tannin ratio of Q. robur foliage, an increase in the pupal weight attained by Operophtera brumata larvae fed on this foliage, but no increase in leaf damage attributed to chewing insects in the field. Berger & Katzensteiner (1994) showed that outbreaks of Pristiphora abietina (Christ) (Hymenoptera: Thentrinidae) (small spruce sawfly) on Picea abies in Austria were associated with high pollution levels, and raised nitrogen and thiol contents of the spruce needles relative to non-outbreak areas.
In heathland systems, there has been an increase in the frequency and severity of Lochmaea suturalis (Thompson) (Coleoptera: Chrysomelidae) (heather beetle) outbreaks on Calluna vulgaris in Holland during the latter half of the present century (see Section 2.3.3). One hypothesis to explain this increase is that deposition of airborne pollutants, in particular ammonia, has caused nitrogen enrichment of heathland vegetation, thereby making Calluna vulgaris more susceptible to outbreaks (Berdowski, 1993). Although there is good evidence that experimental nitrogen-enrichment of C. vulgaris improves L. suturalis larval development rate, survival and adult fecundity (Brunsting & Heil, 1985), there is so far only anecdotal evidence that L. suturalis population densities respond positively to nitrogen enrichment in the field (observations of Blankwaardt, reported in Heil & Diemont, 1983).
It is necessary to be aware of the limitations of studies which attempt to attribute insect outbreaks to pollution regimes. In the past, many such studies have suffered from a purely correlative approach, frequently lacking in controls or replication (Brown, 1995). Given the ubiquitous nature of environmental pollution in industrialised countries, it is difficult to balance the observed incidences of apparently pollution-related outbreaks against the total area of polluted vegetation that is not subject to such outbreaks. More experimental approaches, such as those described by Brown (1995), and studies along pollution gradients, should provide more convincing demonstrations of the effects of pollution on insect populations in the future. Given the strong current evidence for increasing atmospheric nitrogen deposition to British moorlands (Pitcairn et al., 1995), and the conclusion of Brown (1995) that atmospheric pollution by nitrogen dioxide "consistently increases the performance of herbivorous insects", it is important to consider the possibility that the current outbreaks of O. brumata on C. vulgaris are an early response to nitrogen enrichment of Scottish moorlands. This hypothesis is tested and discussed further in Chapter 7.
Further evidence that diet quality can affect insect population densities comes from studies of the temporal relationship between insect emergence and the budburst of their host-plants (Hunter, 1992). The nutritional quality of many plant species declines as their foliage matures (Feeny, 1970; Larsson & Ohmart, 1988), and many insect species, including O. brumata in woodland systems, synchronise their larval emergence with the date of production of the first flush of nutritious young leaves (Nothnagle & Schultz, 1987; Hunter, 1991). The extent to which insect populations successfully achieve this synchrony can affect larval survival and development (Feeny, 1970), the population density achieved by the insect in that year (Holliday, 1977; Thomson et al., 1984), and the overall level of defoliation experienced by the plant (Hunter, 1992; but see Crawley & Akhteruzzaman, 1988). Environmental perturbations of the phenological relationship between insect and host may therefore affect the probability or severity of outbreaks. For example, Dewar & Watt (1992) predicted that future climatic warming will disrupt the synchrony between O. brumata hatching and Picea sitchensis budburst, thereby either reducing the probability of O. brumata outbreaks on this host, or driving the evolution of a compensatory change in O. brumata hatching phenology.
In some cases, different populations of a single insect species show adaptations in their hatching dates which apparently serve to maintain synchrony with the budburst of their respective host populations (Mitter et al., 1979). This divergence can be accompanied by genetic variation between populations (Komatsu & Akimoto, 1995). However, it is necessary to be cautious in the interpretation of observed patterns of hatch-budburst synchrony; the existence of a close temporal relationship between insect emergence and host budburst does not necessarily imply that the former is adapted to the latter, or that the insect gains a nutritional advantage from feeding on young foliage. Myers (1992) experimentally disturbed the close synchrony between the hatching date of Malacosoma californicum pluviale (western tent caterpillar) and the budburst of two of its hosts, Alnus rubra (red alder) and Malus diversifolia (crab apple), and showed that there was no reduction in larval survival or development following a 3 to 6-week hatch delay.
In the present case, the hypothesis that moorland O. brumata populations derive a nutritional and developmental advantage from synchronising their larval emergence with the first growth of C. vulgaris shoots, which are richer in nitrogen than older shoots (Moss, 1967), is tested in Chapter 5, using a similar technique to that of Myers (1992). The existence of such a mechanism would support the hypothesis that C. vulgaris nutritional quality is a major determinant of O. brumata development and outbreak potential in moorland systems.
Although the above discussion (Section 1.2) has shown that increased host quality can elicit positive responses both in individual insect performance and in population density, there are evidently many exceptions to this rule, particularly on conifers (Fig. 1-2). The generality of White's hypothesis that "all kinds of outbreaks" are caused by increased access to nitrogenous food (White, 1993) (Section 1.2.2) is therefore questionable.
An opposing view of insect outbreaks has been developed by Price (1992, 1994), who has stressed the importance of "phylogenetic constraints" which shape the oviposition behaviour and thereby also the emergent population dynamics of a species: latent dynamics are most likely to result from the evolution of highly selective oviposition behaviour (Price et al., 1990), whilst indiscriminate oviposition creates the potential for over-exploitation of local food resources, thereby leading to eruptive dynamics (Price, 1992; Tammaru et al., 1995). Indiscriminate oviposition, by shifting the task of suitable host location to newly-emerged larvae, selects for larvae which are able to disperse easily, and which are highly flexible in their nutritional requirements, able to survive on foliage of extremely low quality (Price, 1992). According to this model, the potential carrying capacity of a forest (or moorland) for an eruptive herbivore is determined only by the total quantity of host foliage present, and consequently "the world really is green for these unusual herbivores" (Price, 1992). Populations of potentially eruptive herbivores must therefore usually be held in check by factors other than host quality (e.g. weather conditions or natural enemies), and outbreaks can be seen as the result of a temporary "release" from these non-nutritional constraints (Price, 1992).
The above model is consistent with the indiscriminate oviposition behaviour of O. brumata (Watt et al., 1992; Graf et al., 1995), and its propensity to outbreak on apparently low-quality hosts such as Calluna vulgaris and Picea sitchensis (see Section 2.2). The following sections therefore review the evidence that factors other than host quality are sometimes responsible for the occurrence of insect outbreaks. Two such potential factors are discussed: climate and natural enemies. Other non-nutritional factors, such as disease (Myers, 1988) and temporal variability in the genetics of eruptive populations (Chitty, 1967; Ruohomäki & Haukioja, 1992a), are outside the scope of the present study, and consequently are not discussed here.
Outbreaks of phytophagous insects have often been linked to episodic climatic events (see Wallner, 1987 for a review). For example, Wellington et al. (1950) found that outbreaks of Choristoneura fumiferana (Clem.) (spruce budworm) in central and eastern Canada were preceded by three or four years of reduced precipitation. Greenbank (1956) subsequently reviewed the available evidence and detailed a theory of "climatic release" to explain C. fumiferana outbreaks, which was later supported by the work of Morris (1963). A similar climatic release mechanism was proposed by Stark (1959a,b) for Recurvaria starki Freeman (lodgepole needle miner) in Canada. Price et al. (1990) pointed out that climatic release mechanisms can be difficult to distinguish from the effects of plant stress and phenology described previously, since meteorological events can affect herbivore performance indirectly via altered host quality, or by changing the phenology of the plant-insect relationship. It is therefore necessary to refine the concept of climatic release in order to distinguish between those climatic effects which act directly on insect populations, and those which act via the host-plants.§
There are some cases in which climate does affect herbivore abundance directly, without any intervening rôle of plant quality. Uvarov (1931, pp 138-140) provided numerous examples of early studies of climatic effects on insect mortality and abundance, and cited the view of Bremer (1928) that Operophtera brumata outbreaks are dependent on winter weather conditions. In the same report, Uvarov (1931, p.138) speculated that "the mortality of insects caused by winter colds is probably the main factor controlling the abundance of most insects in temperate latitudes". More recently, Tenow & Nilssen (1990) showed that the geographical distribution of outbreaks of Epirrita autumnata on birch in northern Fennoscandia is limited by winter temperatures, which regularly fall below the lethal limit to E. autumnata eggs of -37ºC in undamaged forest areas in the Abisko valley and the Finnmarksvidda highland plain (see also Tenow, 1975; Niemelä, 1979). A logical but unproven consequence of this finding is, therefore, that a succession of abnormally warm winters should lead to a "climatic release" of E. autumnata populations in these areas.
Early climatic release theories have, however, received more critical attention in recent years. Royama (1984) re-analysed the data from population studies of Choristoneura fumiferana by Morris (1963), and concluded that cyclic population fluctuations in this species were independent of weather conditions, although weather patterns (and associated migration patterns of egg-carrying moths) were responsible for density-independent secondary fluctuations superimposed upon the primary oscillation. In the same study, Royama proposed that weather events bring independently-oscillating insect populations into synchrony with one another, thereby creating an apparent "climatic release" which might be better termed a "climatic synchronisation" (this mechanism was previously proposed for Canadian lynx populations by Moran (1953)).
Further criticism of climatic release theories has come from Martinat (1987), who drew attention to the lack of statistical rigour of many early studies, and Myers (1988), who argued that "the climatic release hypothesis may be untestable" by standard statistical techniques, due to the difficulty of identifying and measuring the correct climatic variables. There is also a high probability of obtaining spurious significant correlations if large numbers of climatic variables are considered as potential predictors of insect population variability (Myers, 1988). Anecdotal and circumstantial evidence for effects of weather patterns on insect populations should therefore be interpreted with the above caveats in mind. Such observations can lead to the development of interesting hypotheses about the mechanisms governing insect populations (as in this thesis, Chapter 3), but are unlikely to constitute rigorous proofs of such mechanisms.
Natural enemies have been shown to be capable of profoundly affecting the dynamics of insect populations. For example, outbreaks of accidentally-introduced O. brumata in oak forests in Nova Scotia were successfully controlled by the deliberate introduction of two parasitoid species, Cyzenis albicans (Fall.) (Diptera: Tachinidae) and Agrypon flaveolatum (Grav.) (Hymenoptera: Ichneumonidae) (Embree, 1965, 1966; Roland & Embree, 1995). Similar introductions of parasitoids to British Columbia from 1979 to 1981 achieved a decline in O. brumata populations to one-tenth of their peak outbreak density (Roland, 1990). There is evidence that parasitism and pupal predation acted synergistically to bring about the decline in O. brumata population density in British Columbia, since beetle larvae preyed preferentially on unparasitised pupae (Roland, 1990). Subsequent to the decline, O. brumata populations in British Columbia are believed to be stabilised at low levels by density-dependent pupal predation (Roland, 1994, 1995), a conclusion which is in agreement with long-term studies of O. brumata on oak in Britain (Varley et al., 1973). Nevertheless, Roland & Embree (1995) argued that the removal of parasitoids from this system would lead to a resumption by O. brumata of its previous eruptive dynamics.
Density-dependent (and hence potentially regulating) effects of natural enemies on insect populations have been observed in a number of cases (Lessells, 1985; Stiling, 1987), although the ability to detect such phenomena depends upon the spatial scale of the study (Heads & Lawton, 1983; Walde & Murdoch, 1988). If positively density-dependent attack by natural enemies acts with a time lag, cyclic fluctuations may be produced in host population density (Royama, 1977; Myers, 1988). There is some evidence that such an effect contributes to the cyclic dynamics of Epirrita autumnata populations in northern Fennoscandia (Ruohomäki, 1994; but see Bylund, 1995).
Natural enemies play an important rôle in the evolution of host range in phytophagous insects, and the effects of natural enemies can interact with those of plant quality: the colonisation of a new plant species may be facilitated by a reduction in the levels of parasitism and/or predation experienced by herbivore populations on this new host (Brown et al., 1995), and escape from natural enemies can compensate for reduced developmental performance on a poor-quality host (Feder, 1995). Conversely, the presence of natural enemies can prevent the exploitation of high-quality plants: Stiling et al. (1982) showed that high parasitoid abundance prevented effective colonisation of experimentally nitrogen-enriched Spartina alterniflora plants by the leaf miner Hydrellia valida (Diptera: Ephydridae). Increased plant quality may, however, accelerate the rate of development of insect herbivores, thereby reducing their exposure to natural enemies: Loader & Damman (1991) showed that Pieris rapae (Lepidoptera: Pieridae) larvae fed on nitrogen-enriched plants developed more rapidly and experienced lower mortality from predators and parasitoids than larvae fed on low-nitrogen control plants.
In the case of O. brumata on moorlands, it is not yet known how important "top-down" effects of natural enemies are in limiting population densities or geographical distribution. One hypothesis is that moorland O. brumata outbreaks are the result of localised opportunities to escape from regulation by natural enemies, and that exposed Scottish moorlands therefore provide "enemy-free space" for O. brumata (Jeffries & Lawton, 1984; Fryer, 1986). If this hypothesis is correct, then the availability of such enemy-free space will be an important determinant of outbreak distribution. This hypothesis is tested in Chapter 4 and is discussed further in Chapter 8.
The above sections (1.2 & 1.3) highlight a debate about the importance of host-plant nutritional quality for insect development and abundance, and show that in some cases this relationship is apparently weak or absent (Hunter et al., 1991; Waring & Cobb, 1992). One mechanism by which insects can "uncouple" their development from the constraint of host quality is nutritional compensation.
An early study of nutritional compensation was conducted by Slansky & Feeny (1977), who showed that larvae of Pieris rapae responded to low food quality by increasing their rate of feeding. Associated changes were also observed in their efficiency of food use. As a result of this compensatory feeding, P. rapae larvae stabilised their rate of nitrogen uptake, and achieved growth rates that were independent of plant nitrogen concentration (Slansky & Feeny, 1977). Similarly, Paropsis atomaria (Coleoptera: Chrysomelidae) larvae showed compensatory changes in feeding rate in response to manipulation of host quality, and thereby achieved rates of growth and development that were independent of plant nitrogen concentration (above a critical minimum threshold of nitrogen concentration) (Ohmart et al., 1985). Further examples of compensatory feeding by insect larvae were provided by Wheeler & Slansky (1991) and Slansky & Wheeler (1991), and the subject is discussed further in Section 6.1.2.
The hypothesis that O. brumata compensates in the above way for the apparently low nutritional quality of C. vulgaris on Scottish moorlands (Moss, 1967; Iason et al., 1993) is discussed in Section 5.4.2, and is tested experimentally in Chapter 6.
According to the view of Price (1992) (Section 1.3), a polyphagous, eruptive herbivore such as O. brumata should exhibit a high level of nutritional flexibility, perhaps facilitated by the operation of nutritional compensation mechanisms (Section 1.4). However, polyphagy at the species level may alternatively be achieved by dietary specialisation of conspecific populations (or even of individuals within one geographical population) to different hosts (Fox & Morrow, 1981; Akimoto, 1990). Such specialised populations are usually termed "host races" (Jaenike, 1981), and their development is thought to require a restriction of gene flow between populations (Jaenike, 1981; Diehl & Bush, 1984; Fraser, 1995). Specialisation to one host may require a "trade-off", whereby performance on a different host is reduced (Bernays & Graham, 1988; but see Futuyma & Philippi, 1987). The term "biotype" is often loosely used to describe conspecific insect populations which are suspected of having attained a degree of specialisation to different host species, but which have not yet been shown to be distinct host races (but see Fraser (1995) for a critique of this terminology).
Chapter 4 of this thesis addresses the hypothesis that outbreak O. brumata populations feeding on Calluna vulgaris in Scotland constitute a separate host race from broadleaf woodland O. brumata populations. If this is the case, then specialisation of moorland O. brumata populations to C. vulgaris could help to explain the occurrence of outbreaks on this poor-quality host. Falsification of this hypothesis would reinforce the view of Price (1992) (Section 1.3) that eruptive herbivores such as O. brumata are highly nutritionally flexible. In this context, it is interesting to note that a recent Scandinavian study of Epirrita autumnata, which has a similar life history to O. brumata, found no evidence for specialisation to different hosts, either between outbreaking and non-outbreaking E. autumnata populations, or within populations (Ruohomäki & Haukioja, 1992b).
It has been predicted that future environmental changes will produce large-scale responses in vegetation quality, and will elicit major changes in the distribution and abundance of insect herbivores (Bazzaz, 1990; Harrington & Stork, 1995). The predicted doubling in atmospheric carbon dioxide (CO2) concentration by the next century (Houghton et al., 1992) has inspired much recent interest in the effects of CO2 enrichment on plant biochemistry and insect feeding (Lincoln et al., 1993), with a view to predicting such future trends in insect abundance.
Exposure to CO2-enriched atmospheres can have strong effects on plant growth, tissue chemistry and susceptibility to insect herbivores. An increase in photosynthetic rate is usually observed in plants subjected to increased CO2 levels (Lloyd & Farquhar, 1996), and the resultant increase in carbohydrate availability frequently causes a stimulation of plant growth (Bazzaz, 1990; Lloyd & Farquhar, 1996). However, it has been proposed that under conditions of low soil nutrient availability the plant growth response to CO2 can be "nutrient-limited" (Bazzaz, 1990; Woodin et al., 1992); in this proposed condition, it is not possible for the plant to utilise the whole of its increased carbohydrate production for growth, because of lack of nutrients. Surplus carbohydrate will then "dilute" plant tissue nitrogen content, thereby reducing the nutritional quality of the plant for herbivores (Lincoln, 1993). Furthermore, the carbon/nutrient balance hypothesis (Bryant et al., 1983) predicts that this increased internal carbon availability will trigger an increase in the synthesis of carbon-based secondary defence chemicals (Lincoln, 1993), which will further reduce plant quality for herbivores (Feeny, 1976).
The proposed interaction between plant responses to CO2 enrichment and nutrient availability described above is, however, difficult to demonstrate. Nutrient limitation of plant growth response to CO2 enrichment, as proposed above, was described by Woodin et al. (1992) in Calluna vulgaris. However, Lloyd & Farquhar (1996) reviewed the current literature and concluded that there was little experimental evidence that nitrogen-limitation of plant growth responses to CO2 enrichment is a common phenomenon (see Fig. 1-3).
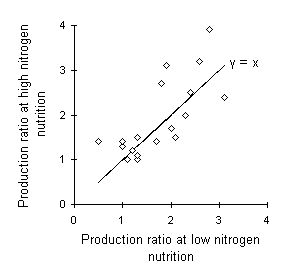
Figure 1-3. The effects of CO2 enrichment on 16 different plant species under both low and high nitrogen availability in 12 different studies. Production ratio is calculated as (rate of biomass accumulation at twice-ambient [CO2]) / (rate of biomass accumulation at ambient [CO2]). Solid line shows equality between low-nitrogen and high-nitrogen responses. There is no evidence that the production ratios differ significantly between nitrogen treatments (t17 = 1.18, n.s.). Data obtained from Table 1 in Lloyd & Farquhar (1996).
Many studies have failed to demonstrate the positive effect of CO2 enrichment on plant carbon-based secondary compound production which is predicted by the carbon/nutrient balance hypothesis (for a review, see Lincoln et al., 1993). However, Lindroth et al. (1993) showed that levels of condensed and hydrolysable tannins were significantly increased by exposure to raised CO2 concentrations in one tree species out of the three investigated: Acer saccharum (sugar maple). Lavola & Julkunen-Tiitto (1994) provided evidence for interacting effects of CO2 enrichment and NPK fertilisation on soluble carbohydrate pools and levels of phenolic secondary compounds in Betula pendula, and these responses were mostly in agreement with the predictions of the carbon/nutrient balance hypothesis.‡ Similarly, Roth & Lindroth (1994) showed an increase in condensed tannin content and a drop in foliar nitrogen concentration following CO2 enrichment in Betula papyrifera (paper birch), but not in Pinus strobus (white pine).
Given the complexity of the above plant responses, it is not surprising that the resultant effects on insect herbivory are also variable. Lindroth et al. (1993) showed that different insect species responded in different ways to CO2 enrichment of a single host species. Watt et al. (1995) reviewed the 15 studies of the effects of CO2 enrichment on 19 different insect-plant interactions published between 1984 and 1993, and drew attention both to the diversity of responses in insect growth and development obtained, and to the small proportion of cases in which those responses were statistically significant. In those cases where significant effects of host CO2 enrichment on insect growth and development were found, they were usually negative (Watt et al., 1995). There was, however, clear agreement between studies that CO2 enrichment caused a reduction in plant tissue nitrogen concentration, and there was also strong evidence that many insect herbivores compensated for this reduction by increasing their consumption rate, as described above (Section 1.4) (Watt et al., 1995; see also Lincoln et al., 1993). The latter result suggests that the long-term effects of rising CO2 levels on insect herbivore dynamics will depend on the ability of the insect species in question to compensate for changes in host quality (Section 1.4).
With the above arguments in mind, Chapter 7 investigates the effects of increased CO2 concentrations on the interaction between O. brumata and C. vulgaris. In view of the possibility that nutrient limitation may restrict the response of C. vulgaris to CO2 (Woodin et al., 1992), the effects of nutrient enrichment on the system are investigated in combination with those of CO2 (Chapter 7).
Chapter 2 provides a detailed introduction to the phenomenon under study, O. brumata outbreaks on C. vulgaris-dominated moorlands. The study organisms and study sites are described, and the effects of insect outbreaks on moorland vegetation are reviewed.
Before commencing manipulative studies of an insect-plant system, it is important to possess a basic knowledge of its ecology in situ. Chapter 3 therefore focuses on the patterns of resource utilisation by O. brumata in moorland sites. Larval and adult population densities in the field are measured. Development of O. brumata on C. vulgaris is compared with that on Vaccinium myrtillus (L.) (Ericaceae), in order to test the hypothesis that C. vulgaris is a low-quality host. Evidence is presented that winter weather patterns have a strong influence on O. brumata population levels on high-altitude moorlands in mainland Scotland, and that outbreaks therefore require a "climatic release" (see Section 1.3.2).
Chapter 4 tests the hypothesis that moorland O. brumata populations are specialised to feeding on moorland plants (Section 1.5). Differences in life history and pupal respiration rate are demonstrated between moorland and non-moorland populations of O. brumata, but it is argued that these differences are more closely linked to altitude than to specialisation on moorland vegetation. A reciprocal transfer experiment provides no evidence to support the hypothesis that moorland and oak-feeding O. brumata populations are specialised to C. vulgaris and Quercus robur respectively. Parasitism rates in different O. brumata populations are also compared, and it is shown that high-altitude moorlands can provide "enemy-free space" (Section 1.3.3) for O. brumata.
Chapter 5 focuses on differences in hatching phenology between moorland and oak-feeding O. brumata populations, and refutes the hypothesis that synchrony between larval emergence and budburst of Calluna† is important for O. brumata survival and development in moorland systems (see Section 1.2.4). The rôle of nutritional compensation (Section 1.4) in allowing O. brumata to feed on poor-quality hosts such as Calluna is discussed, and is then tested experimentally in Chapter 6.
Chapter 7 evaluates the effects of increased atmospheric CO2 concentration and increased nitrogen availability on the O. brumata-C. vulgaris interaction (see Section 1.6), and finds no evidence to support the hypothesis that nitrogen-enrichment of moorland vegetation (Sections 1.2.3 & 2.3.3) is the cause of the current O. brumata outbreaks.
Chapter 8 concludes the thesis by reviewing the results of Chapters 3 to 7 in the light of the above debate about the causes of insect outbreaks (Sections 1.2 and 1.3). The evidence that host quality is an important determinant of O. brumata performance in moorland systems is evaluated critically, and the hypothesis that moorland O. brumata outbreaks are caused by localised host-plant enrichment is discussed. Alternative hypotheses concerning the potential rôles of weather patterns and natural enemies in allowing the "release" of moorland O. brumata populations (Section 1.3) are considered. The relative importance of "top-down" versus "bottom-up" effects (Hunter & Price, 1992) on moorland O. brumata populations is discussed. The potential effects of future environmental changes on the system are considered, with the aid of a "functional scheme" for such predictions devised by Landsberg & Smith (1992).
† But note that these feeding trials were conducted using foliage excised from the host-plant: for potential disadvantages of this approach, see Risch (1985).
§ Examples of indirect effects of weather on insect populations, apparently acting via host-plant quality, are discussed in Section 1.2.2, whilst climatic effects on insect-plant phenological synchrony are discussed in Section 1.2.4.
‡ although, regrettably, the degrees of freedom were omitted from the published statistical tables in this study, and it is therefore impossible to determine whether a pseudoreplicated form of analysis was employed (see Hurlbert, 1984; Fowler & Lawton 1985).
† For ease of reading, Calluna vulgaris may be referred to simply as "Calluna" throughout the text.
 Contents Page |
Next Chapter |