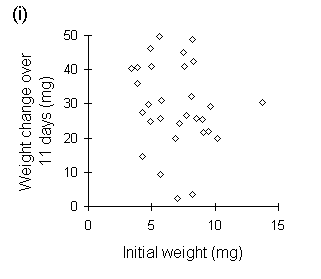
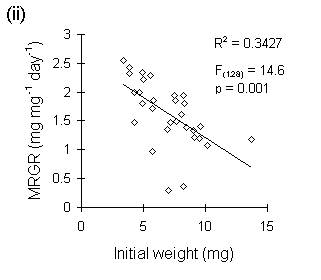
Figure I-3. Larval growth data from reciprocal transfer experiment
in Section 4.3.4. (i): lack of relationship between larval growth over
the trial and initial larval weight; (ii): negative relationship between
MRGR and initial larval weight.
This appendix provides a rationale for the methods used to calculate larval growth rates in Chapters 3 to 7. Problems with one common index of growth rate, the mean relative growth rate (MRGR) (Radford, 1967) are highlighted.
Because the growth of Lepidopteran larvae does not proceed linearly with time, but is approximately proportional to existing body weight (Gordon, 1968), larval growth rate in feeding trials is commonly expressed as a mean relative growth rate (Radford, 1967), calculated as follows:
MRGR = [ln(final weight) - ln(initial weight)] / length of feeding period
expressed in mg mg-1 day-1.
However, the application of the above formula becomes problematic if, as in this thesis, feeding trials begin at the egg stage or with low-weight early instar larvae. Under these circumstances, ln(initial weight) becomes a large negative number, which results in anomalously high MRGR values and a spuriously high sensitivity of this growth index to small variations in initial weight.
This problem is illustrated in Figure I-1. A simulated pattern of O. brumata growth is shown (Fig. I-1i), based on data from observations of O. brumata development on C. vulgaris made during the course of this study. The logarithm of this body weight data is plotted against time in Figure I-1ii, and the gradient of the resultant line constitutes the MRGR over any given feeding period.
Figure I-1.
Simulated growth pattern for O. brumata on C. vulgaris:
(i): untransformed fresh weight against time;
(ii): log-transformed fresh weight against time.
Whilst the logarithmic transformation achieves partial linearisation of the O. brumata growth curve, there is distortion away from linearity at the early stages of larval development. Consequently, MRGR values calculated over feeding periods which commence at these early larval stages will be biased upwards and incomparable with values which are measured only over later larval instars. This is shown in Figure I-2, which plots the MRGR values for hypothetical 16-day and 28-day feeding trials, calculated from the simulated body weight data given in Figure I-1i, against the starting date of these feeding trials.
Figure I-2.
Mean relative growth rates for hypothetical O. brumata feeding trials over
(i): 16 days and (ii):
28 days, plotted against date of initiation of trial.
MRGR values are calculated from the simulated
O. brumata growth curve given in Figure I-1.
The logarithmic MRGR values shown in Fig. I-2 vary by a factor of three, depending on the precise stage of larval development measured. As expected, logarithmic MRGR values are displaced upwards for feeding trials commencing with early-instar larvae (Fig. I-2i , days 1-5).
These results reinforce the need for standardisation of methodologies between different studies before their results can be compared meaningfully. Measurements of larval relative growth rates taken over different stages of larval development can clearly be expected to differ substantially (e.g. Watt et al., 1996).
A second, closely related problem with the use of MRGR arises from the assumption that growth is proportional to body weight. Despite Radford's (1967) argument that this assumption is not necessary for the use of MRGR, its violation in limited-duration larval feeding trials can produce artefactual results, even if such trials are restricted to the later instar stages. If larval growth over the course of the trial is not proportional to weight at the start of the trial (Fig. I-3i), then an artefactual negative relationship arises between MRGR and initial weight (Fig. I-3ii); larvae which are relatively small at the start of the feeding trial achieve large MRGR values, simply by virtue of their low starting weight.


Figure I-3. Larval growth data from reciprocal transfer experiment
in Section 4.3.4. (i): lack of relationship between larval growth over
the trial and initial larval weight; (ii): negative relationship between
MRGR and initial larval weight.
The above drawbacks of MRGR highlight the potential danger of its indiscriminate use (see also Packard & Boardman (1988) and Raubenheimer (1995) for discussion of the problems of ratio-based growth indices). It is therefore necessary to consider carefully whether MRGR is the most appropriate index of larval growth for use in larval feeding trials.
The approach taken in this thesis has been to abandon the use of MRGR altogether, for the reasons above, and whenever possible to perform statistical analyses on simple untransformed larval growth rates (mg day-1) (Chapters 3 & 5). On two occasions, however, a square root transformation was applied to larval weight data prior to the calculation of growth rates (Chapters 6 & 7), in order to fulfil the assumptions for analysis of variance (Mead et al., 1993). The use of a square root transformation does not incur the same problems as discussed above for the MRGR: there is less distortion at low weights (Fig. I-4), and lower probability of an artefactual relationship between growth rate and initial larval weight (Fig. I-5).
Figure I-4. Square
root transformation of the simulated O. brumata growth data given in Figure
I-1i.
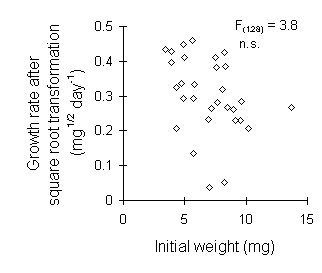
Figure I-5. Lack of relationship between growth rate after square
root transformation and initial larval weight for the reciprocal transfer
data shown in Figure I-3.
Square root transformation proved highly effective at linearising larval
growth responses in the feeding trial described in Chapter 6 (Fig. I-6),
and resulted in slightly higer R2 values than logarithmic transformation
(Fig. I-7).
Figure I-6. (i) & (ii):
Patterns of individual O. brumata larval weight increase on C. vulgaris
in the feeding trial in Chapter 6. (iii) &
(iv): linearisation of larval growth patterns
from (i) & (ii) (excluding final points of falling weight), using square
root and logarithmic transformations.
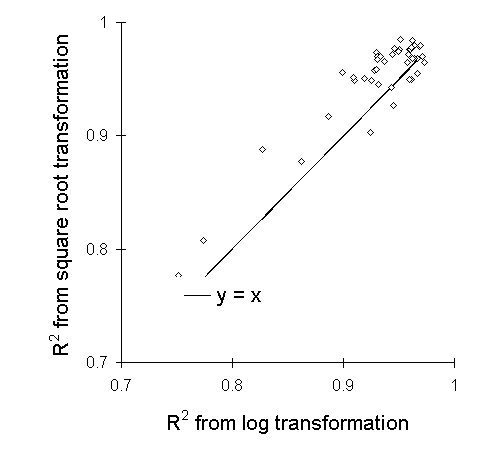
Figure I-7. R2 values resulting
from linearisations of individual larval growth responses by square root
and logarithmic transformations. Solid line indicates equality between
the two transformations. Square root transformation achieves slightly higher
R2 values than logarithmic transformation
in most cases.
This appendix compares the values of plant total nitrogen concentration obtained from two different assay methods: acid digestion (Cresser & Parsons, 1979) and combustion in a NCS autoanalyser (see Sections 7.2.1 & 7.4.5).
A total of 10 samples of Nardus stricta foliage and 5 samples of Calluna vulgaris foliage were assayed for total nitrogen concentration by both acid digest and NCS autoanalyser techniques. The samples were washed with deionised water, dried for three days at 80ºC and ground prior to analysis.
For the acid digest technique †, samples were digested in a mixture of perchloric and concentrated sulphuric acids for 90 minutes at 365ºC, then diluted with deionised water (see Cresser & Parsons (1979) for the full digest methodology). Samples were then assayed for ammonium-N colorimetrically, by the indophenol-blue reaction, using a "Technicon" Autoanalyser II (Serlin Instruments, Herts, U.K.).
For the NCS assay, the ground samples were assayed for total nitrogen using a Fisons NA 1500 autoanalyser, calibrated with a bulked soil standard (as in Section 7.2.1).
The NCS analyser gave consistently higher results than the acid digest technique (Fig. II-1). NCS values were greater than acid digest values by a mean factor of 11.6% (s.e. = 1.62%). The mean difference in total nitrogen concentrations between assay methods was 0.184% N (s.e. = 0.027% N). Despite these differences, there was a strong curvilinear relationship between the two sets of values (Fig. II-1).
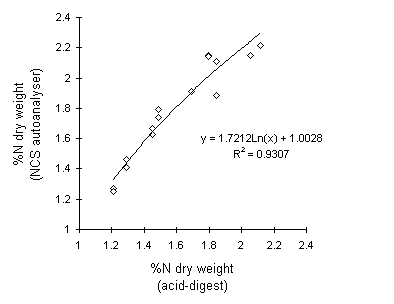
Figure II-1. Relationship between plant total nitrogen concentrations
(as a percentage of dry weight) obtained by NCS and acid digest assay methods.
The implications of the above data for the Calluna tissue nitrogen measurements in this study are discussed in Section 7.4.5. The results of this study demonstrate the need to consider assay methodology, as well as sampling date (Section 7.4.5), when interpreting published plant tissue nitrogen data. Attempts to detect nitrogen-enrichment of plants in the field must be particularly careful to control for variability from these sources.
† These acid digestions and colorimetric assays were performed by W.K. Hicks.